Psychological Distress, Health Behaviors, and Benefit Finding in Survivors of Multiple Primary Cancers: Results From the 2010 Livestrong Survey
Purpose/Objectives: To evaluate whether survivorship of multiple primary cancers (MPCs) is associated with psychological distress, positive health behaviors, and benefit finding.
Design: Secondary analysis of the 2010 Livestrong cross-sectional survey.
Setting: Online survey.
Sample: 238 MPC survivors and 3,295 single cancer survivors.
Methods: Chi-square and t tests for group comparisons were used. Multivariate linear regression, adjusted for covariates, was used to determine associations between variables.
Main Research Variables: MPC versus single cancer; psychological distress, health behavior (healthy lifestyle and positive healthcare utilization), and benefit-finding scores.
Findings: Survivors of MPCs (compared to single cancer survivors) were significantly older, less likely to have a spouse or partner, further out from original cancer diagnosis, and less likely to be employed full-time, and they differed by cancer diagnoses and survivorship stage. Having MPCs was associated with significantly higher psychological distress and healthcare utilization but not healthy lifestyle or benefit finding.
Conclusions: Relative to those with single cancers, MPC survivors are at increased risk for psychological distress and are more likely to receive recommended cancer screenings. Additional research is needed to understand mechanisms surrounding psychological distress in MPC survivors.
Implications for Nursing: Targeted distress screening in MPC survivors may allow for early identification and interventions to ameliorate distress and reduce negative downstream health effects.
Jump to a section
One in five cancers diagnosed in the United States will occur in someone who has a previous cancer diagnosis, and these multiple primary cancers (MPCs) are a major cause of morbidity and mortality in cancer survivors (Bluethmann, Mariotto, & Rowland, 2016; De Gonzalez et al., 2011; Morton, Onel, Curtis, Hungate, & Armstrong, 2014).
A second cancer, or MPC, is the occurrence of a new cancer that is histologically distinct from the original primary cancer and has been ruled out as metastatic disease of the primary tumor (Begg, 1999). An example of someone who is an MPC survivor is an individual who experiences breast cancer and later presents with a new diagnosis of ovarian cancer. Contrast this with a woman with breast cancer that metastasizes to the bone, which is diagnosed as metastatic spread of the original breast cancer; this would not be considered an MPC. Risk of developing subsequent MPCs varies by site of first primary cancer, age at first cancer diagnosis, environmental and behavioral exposures, genetic susceptibility, and cancer treatment effects (American Cancer Society, 2009, 2014; Morton et al., 2014).
The National Academy of Medicine, other professional organizations, cancer survivorship advocates, clinicians, and scientists have called for an increased focus on addressing the health and psychosocial needs of the growing population of cancer survivors (American Cancer Society, 2016; Institute of Medicine, 2006; Klein et al., 2014; Knobf et al., 2015; Miller et al., 2016; Mullan, 1985, 2016), and the MPC population represents an understudied and at-risk group in critical need of additional research. Although having a single cancer has been linked to risks for psychological distress (Holland et al., 2013; Mitchell et al., 2011; National Comprehensive Cancer Network, 2017a), poor health behaviors (Mowls, Brame, Martinez, & Beebe, 2016; Underwood et al., 2012), and poor physical health outcomes (Ness, Wall, Oakes, Robison, & Gurney, 2006; Stein, Syrjala, & Andrykowski, 2008) that can persist throughout cancer survivorship, an initial small body of literature is evolving to suggest that the risk for these poor outcomes appears to be even greater in MPC survivors (Andrykowski, 2012; Belcher, Hausmann, Cohen, Donovan, & Schlenk, 2016; Burris & Andrykowski, 2011; Dowling et al., 2013; Gotay, Ransom, & Pagano, 2007; Thong et al., 2013). Most cancer survivorship literature, however, has been conducted irrespective of the number of cancer diagnoses, limiting the ability to understand potentially unique experiences and needs in this survivor subset. In addition, no studies of MPC survivors have analyzed a large national dataset, such as the Livestrong survey, that focuses entirely on post-treatment cancer survivorship issues.
Many cancer survivors experience persistent late and/or long-term effects of cancer and cancer treatment (National Comprehensive Cancer Network, 2017b). Uncontrolled psychological distress in cancer survivors is known to negatively affect quality of life, adherence to surveillance recommendations, and engagement in health-promotion activities (National Comprehensive Cancer Network, 2017a). Previous cancer survivorship literature has demonstrated that healthy lifestyle behaviors are associated with decreased chronic illness and improved health and quality of life (Blanchard, Courneya, & Stein, 2008; Davies, Batehup, & Thomas, 2011; Ford et al., 2009). Benefit finding, the perception of positive changes (e.g., renewed appreciation for life following adversity), has been found in single cancer populations and may also be related to positive health behavior change and psychological adjustment (Harper et al., 2007; Hawkins et al., 2010; Kanera et al., 2016; Low et al., 2014). Previous cancer survivorship research has been conducted without consideration of patients’ history of MPCs, but early evidence suggests that this growing population of MPC survivors may be at an increased health risk, highlighting a critical need to build the science to identify potentially modifiable risk and protective factors contributing to health outcomes in this unique cancer survivor population.
The purpose of this secondary analysis of 2010 Livestrong national cancer survivorship survey data is to evaluate whether MPC survivorship is associated with psychological distress, positive health behaviors, and benefit finding. The current article reports (a) sociodemographic and clinical differences between survivors of single cancers versus MPCs and (b) the contribution of MPC survivorship to psychological distress, positive health behaviors, and benefit finding after controlling for important covariates. Findings from this study were used to make recommendations applicable to a wide range of nurses to support MPC survivors.
Methods
The 2010 Livestrong Survey for People Affected by Cancer was a cross-sectional survey conducted online from June 2010 to March 2011. Constituents of Livestrong were notified about the survey via email, Twitter, and Facebook. Partner organizations, state cancer coalitions, and comprehensive cancer centers shared survey information with their respective constituents and/or patients (Beckjord et al., 2014; Campbell et al., 2011; Shapiro et al., 2009).
The 2010 Livestrong survey was developed in response to recommendations from the Institute of Medicine (2006), now referred to as the Health and Medicine Division of the National Academies of Sciences, Engineering, and Medicine, that nonprofit organizations increase their support of cancer survivorship research and associated mechanisms and was aimed at examining post-treatment survivorship issues. The Livestrong Foundation developed items for the preceding 2006 Livestrong survey through a multiyear formative research process, during which experts and cancer survivors were consulted to incorporate challenges faced by cancer survivors. Many of the 2006 Livestrong survey items were retained in the 2010 survey following a RAND Corporation analysis that examined survey response patterns and content (Rechis et al., 2011). Main topic areas in the 2010 survey included physical, emotional, and day-to-day concerns, as well as meaning making, information seeking, advocacy, and engagement. Additional details regarding survey development, participant recruitment, and survey administration have been previously published (Beckjord et al., 2014; Low et al., 2014; Posluszny et al., 2015; Rechis et al., 2011).
Sample
The parent study received institutional review board (IRB) approval (Rechis et al., 2011), and this analysis of deidentified 2010 Livestrong survey data was approved by the University of Pittsburgh IRB. The initial survey included 4,192 post-treatment adult cancer survivors whose data were considered for this study. Sample selection by single and MPC groups is described here and presented as a flow chart in Figure 1.

Survey respondents were asked to report their type of cancer (primary site) and could choose from an extensive 88-item checklist of cancer types. Respondents were also asked to separately identify any additional cancer diagnoses or recurrences. A priori decisions were made to exclude nonmelanoma skin cancer cases in single and MPC groups. Additional exclusion criteria for the MPC group included second cancer identical to first primary cancer (i.e., recurrence); definite or probable metastatic disease for common sites of cancer metastases (i.e., bone, liver, lung, and brain) (National Cancer Institute, 2017); and/or unclear, missing, “I don’t know,” or non-cancer “other” diagnoses that were not actual cancer diagnoses (e.g., stroke).
Variables of Interest
Respondents were classified as either single or MPC survivors as previously described. The following categories of variables were assessed in the Livestrong survey by asking, “Since completing treatment, have any of the following statements been true for you as a result of your experience with cancer?” A series of statements followed, to which respondents could answer “yes,” “no,” or “I don’t know.” “I don’t know” replies were treated as missing data in this study. Individual survey item endorsements were used to compute sum scores for the four survey outcome categories of interest.
Psychological distress, healthy lifestyle, and benefit finding: Psychological distress consisted of eight items pertaining to anxiety; worry, tension, or stress; preoccupation with cancer; worry about dying from cancer; worry about cancer recurrence; depression; and mood swings. Health behaviors were divided into two categories: healthy lifestyle and positive healthcare utilization. The four healthy lifestyle behavior items were leading a healthier lifestyle, regular physical activity (two to three times per week), healthier diet, and attempts to take care of health. Positive healthcare utilization included three items: attending regular medical appointments, monitoring for second cancer, and being up to date on recommended cancer screenings. Six benefit-finding items included greater appreciation for life, recognition of what’s important in life, renewed spirituality, ability to better deal with stress, better coping, and overall feeling like a better person.
Sociodemographic and clinical variables: Sociodemographic variables consisted of age at survey, gender, race, partner status (i.e., single, divorced/widowed, or married), children younger than age 18 years living in the home, educational status, total household income, and employment status. Cancer-related clinical variables included age at initial cancer diagnosis, years since diagnosis, first primary cancer diagnosis (included categories for top five most prevalent diagnoses represented by respondents [breast, testicular, colorectal, hematologic, and prostate]; remaining diagnoses were represented by “other”), years since last treatment, stage of survivorship (i.e., currently on treatment, living with cancer as a chronic illness, less than one year post-treatment, one to five years post-treatment, greater than five years post-treatment, and prefer not to answer/unsure), and cancer treatment received (i.e., no chemotherapy, chemotherapy only, or chemotherapy plus surgery and/or radiation). The selection of these predictor variables was driven by critical variables identified in the MPC literature (Andrykowski, 2012; Belcher et al., 2015, 2016; Burris & Andrykowski, 2011; Dowling et al., 2013; Thong et al., 2013).
Analyses
Descriptive statistics were used to characterize the sample and key variables of interest. To compare characteristics between single cancer and MPC groups, the authors used independent sample t tests for continuous variables and chi-square for categorical variables. Post-hoc contingency table analyses using Pearson’s chi-square test were conducted for categorical variables reaching statistical significance, and Bonferroni adjusted p values were calculated to correct for type I error.
Predictor variables of interest were selected a priori based on the literature and were included in the empirically driven multivariate analyses. Multivariate linear regression analysis with listwise deletion was used to develop models for predicting the overall categories of psychological distress, healthy lifestyle behaviors, positive healthcare utilization, and benefit finding, adjusted for statistically (p < 0.05) and theoretically significant covariates. Variables were included as model covariates if they (a) were related to MPC in bivariate analyses at p < 0.05 or (b) were associated with outcomes in previously published work (i.e., were statistically or theoretically significant). Predictor variables included the following sociodemographic and clinical variables: age at time of survey and at initial diagnosis; time since first diagnosis; gender; race; marital status; children younger than age 18 years living in the home; education; income; employment status; first primary cancer diagnosis; time since last treatment; stage of survivorship; type of cancer treatment(s); and survivorship of MPC. Test statistics are presented for each full regression model followed by standardized beta and p values for statistically significant predictors within each model.
The data were analyzed using SPSS®, version 22. All tests were two-tailed, and the statistical significance criterion threshold was set at p < 0.05 unless otherwise noted for Bonferroni corrections.
Results
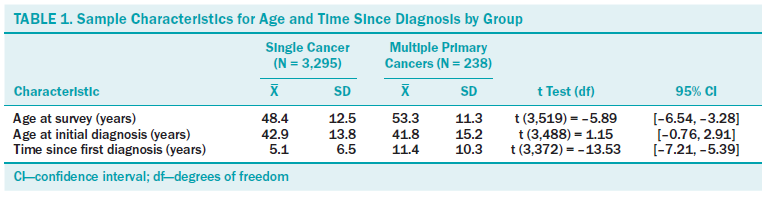
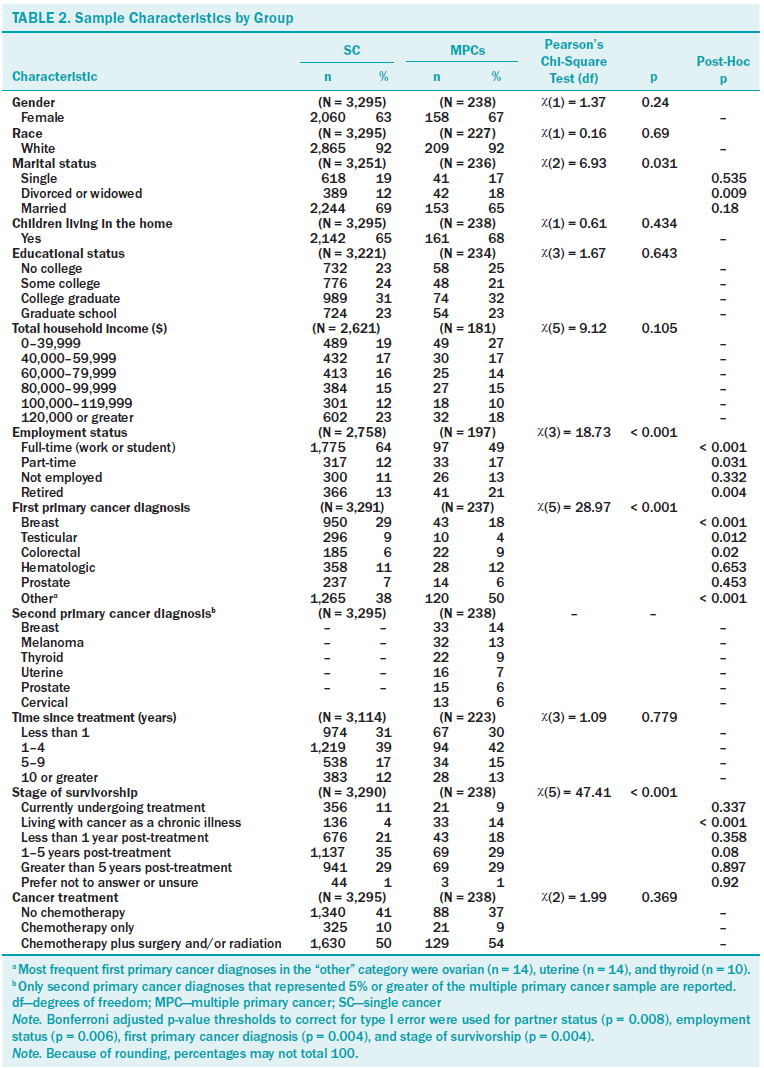
Descriptive statistics are displayed in Tables 1 and 2 for single cancer (N = 3,295) and MPCs (N = 238) groups. MPC participants differed significantly from those with single cancer diagnoses in that they were older at the time of survey completion and were further out from their initial diagnosis. In addition, groups differed statistically by partner status, employment status, type of first primary cancer diagnosis, and stage of survivorship. Specifically, those with MPCs were less likely to have had breast cancer and were more likely to have had one of the less common cancers represented in the dataset (i.e., “other”) as a first primary cancer diagnosis. First primary cancer diagnoses most frequently represented in the “other” category for MPC survivors included ovarian, uterine, and thyroid cancers. Breast cancer and melanoma were the two most commonly reported second primary cancer diagnoses for MPC survivors. MPC survivors were also more likely than single cancer survivors to endorse living with cancer as a chronic illness when identifying their stage of survivorship. Being divorced or widowed was more common in MPC survivors, but this difference was not significant after Bonferroni adjustment.
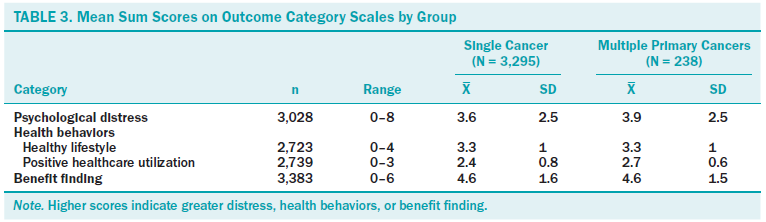
Mean scores for primary outcomes by single and MPC groups are displayed in Table 3. The final psychological distress model accounted for 8% of the model variance (F[35, 2,670] = 7.51, p < 0.001). Significant predictors of psychological distress in the final model included age at survey (standardized beta = –0.195, p = 0.012), gender (female [standardized beta = 0.171, p < 0.001]), partner status (divorced or widowed [standardized beta = 0.075, p = 0.002] and married [standardized beta = 0.054, p = 0.045]), first primary cancer diagnosis (colorectal [standardized beta = 0.042, p = 0.045]), stage of survivorship (living with cancer as a chronic illness [standardized beta = 0.057, p = 0.006]), and survivorship of MPCs (standardized beta = –0.021, p = 0.021).
The final healthy lifestyle behaviors model accounted for 1% of the model variance (F[35, 2,378] = 1.88, p = 0.001. Significant predictors of healthy lifestyle in the final model included race (non-White [standardized beta = 0.057, p = 0.006]), total household income ($80,000–$99,999 [standardized beta = 0.05, p = 0.031] and $100,000–$119,999 [standardized beta = 0.06, p = 0.009]), employment status (not employed [standardized beta = –0.053, p = 0.013]), time since last treatment (five to nine years [standardized beta = –0.082, p = 0.011]), and stage of survivorship (greater than five years post-treatment [standardized beta = 0.086, p = 0.029]).
The final positive healthcare utilization behaviors model accounted for 4% of the variance in healthcare utilization (F[35, 2,392] = 3.8, p < 0.001). Significant predictors of healthcare utilization in the final model included educational status (college graduate [standardized beta = 0.068, p = 0.009]), first primary cancer diagnosis (colorectal [standardized beta = 0.053, p = 0.021] and prostate [standardized beta = –0.065, p = 0.017]), cancer treatment (chemotherapy plus surgery and/or radiation [standardized beta = 0.047, p = 0.046]), and survivorship of MPCs (standardized beta = 2.899, p = 0.004).
The final benefit-finding model accounted for 3% of the model variance (F[35, 2,958] = 3.38, p < 0.001). Significant predictors of benefit finding in the final model included race (non-White [standardized beta = 0.053, p = 0.003]), partner status (divorced or widowed [standardized beta = –0.051, p = 0.03] and married [standardized beta = –0.055, p = 0.033]), having children younger than age 18 years living in the home (standardized beta = 0.114, p < 0.001), total household income ($100,000–$119,999 [standardized beta = 0.043, p = 0.035]), employment status (not employed [standardized beta = –0.061, p = 0.001]), and cancer treatment (chemotherapy only [standardized beta = 0.056, p = 0.007] and chemotherapy plus surgery and/or radiation [standardized beta = 0.046, p = 0.029]).
Survivorship of MPCs, the primary predictor variable of interest, was significantly associated with psychological distress (standardized beta = 0.046, p = 0.021) and positive healthcare utilization behavior models (standardized beta = 2.899, p = 0.004) but not with healthy lifestyle behaviors (standardized beta = –0.012, p = 0.585) or benefit finding (standardized beta = 0.011, p = 0.562).
Discussion
Most striking in this study was the association between MPC diagnoses and psychological distress, which was consistent with the authors’ review of the literature (Belcher et al., 2016). Consistent with findings in a cohort of MPC survivors 10–20 years older than the MPC survivors in the current sample (Gotay et al., 2007; Thong et al., 2013), survivorship of MPCs did not predict benefit finding as a result of one’s cancer experience. An unexpected finding unique to this study was that MPC survivors were more likely than single cancer survivors to report “living with cancer as a chronic illness” when asked to identify their stage of survivorship. This finding may indicate that MPC survivors face additional survivorship needs related to chronic illness and warrants additional study. Living in a state of chronic illness may be contributing to chronic stress and increasing risk for physical and psychological disease in this population (Corbin & Strauss, 1988; Dowrick, Dixon-Woods, Holman, & Weinman, 2005; Grady & Gough, 2014; Miller, Cohen, & Ritchey, 2002).
Consistent with other MPC studies, the authors found that MPC survivors differed from single cancer survivors in that they were older (Andrykowski, 2012; Thong et al., 2013) and were further out from their initial cancer diagnosis (Burris & Andrykowski, 2011). However, the MPC survivors represented by this Livestrong cancer survivor sample were, on average, about 11–18 years younger than those represented in previous MPC literature (Andrykowski, 2012; Burris & Andrykowski, 2011; Gotay et al., 2007; Thong et al., 2013). In addition, MPC and single cancer survivors in this sample also differed by type of initial cancer diagnosis, with MPC survivors being less likely to have had breast cancer as their first diagnosis and more likely to fit into the “other” category (i.e., ovarian, uterine, and thyroid). With differing cancer types come differing treatments and cancer treatment experiences. Therefore, additional research is needed to determine the complex implications of differing diagnoses and treatments on health outcomes in MPC survivors.
Although this study did not find statistical differences for income between groups, MPC survivors were less likely to be employed full time and more likely to be retired. Other preliminary work by the current authors has found that MPC survivors with recurrent ovarian cancer were more likely to endorse lower income and difficulty meeting basic needs than survivors with recurrent ovarian cancer only (Belcher et al., 2015). Another study found that MPC survivors experienced greater levels of lost productivity (e.g., employment) as compared to individuals without cancer and to survivors of single cancers (Dowling et al., 2013). With respect to partner status, the authors found that being divorced or widowed was more common in MPC survivors, but post-hoc testing with Bonferroni adjustments for type I error did not identify statistical differences. Partner status (i.e., divorced or widowed and married) was predictive of psychological distress. A study from the Netherlands found that MPC survivors reported greater cancer impact on life, including body changes and interference with social activities (Thong et al., 2013). When viewed in context with findings from previous studies, results from this study support further examination of the effect of MPC on work and social role function in future MPC studies.
MPC survivors were more likely to report positive healthcare utilization, including engagement in cancer screenings and regular medical appointments. Similarly, Thong et al. (2013) found that MPC presence was associated with greater health awareness. Conversely, MPC status was not associated with healthy lifestyle behaviors, such as diet and regular exercise, which was consistent with Burris and Andrykowski’s (2011) findings that those with MPCs were more likely than single cancer survivors to have unhealthy behaviors (i.e., physical inactivity, smoking, and alcohol use). This may reflect a maladaptive behavioral coping response and warrants additional study in MPC survivors. As day-to-day chronic disease management responsibility shifts from providers to individuals (Barlow, Wright, Sheasby, Turner, & Hainsworth, 2002; Ryan, 2009), interventions to support survivors in initiating and maintaining healthy behaviors will be increasingly important in limiting exacerbation of existing conditions and preventing new conditions.
Limitations and Strengths
Given the cross-sectional design, causal or temporal relationships between variables cannot be determined. Secondary analysis is limited to questions posed in the dataset, and information about psychological distress severity was not collected. Although the authors could account for 8% of variance in psychological distress in this large sample of cancer survivors, this suggests that other important factors exist that were not able to be included in this secondary data analysis, such as comorbidities, symptoms, physical function, perceived stress, social support and coping resources, self-management behaviors, financial toxicity, and biologic stress responses. In addition, MPC survivors represented just 5.7% of the sample, which is slightly less than the 8% MPC representation that is typically found in the overall cancer survivor population (Mariotto, Rowland, Ries, Scoppa, & Feuer, 2007). By conservatively excluding cases in which survivors reported a common site of metastasis as their second cancer, it is possible that the authors may have excluded true MPC cases from the analyses. In addition, it has previously been reported that Livestrong respondents are younger, less diverse, more educated, and wealthier than would be expected, which may be because of the voluntary, online nature of this survey (Low et al., 2014; Rechis et al., 2011) and may lead to decreased generalizability to the general cancer survivor population. However, this study expands what is currently known about MPC survivors by capturing a sample of survivors at an earlier age than has previously been described. Lastly, missing data, mostly in health behavior outcomes, may bias findings. Because rates of missingness were similar for variables between groups, the authors included as many cases as possible for both groups and presented all available data.
Strengths of this study include the ability to capture a large sample of post-treatment MPC survivors, to provide data on a younger demographic of MPC survivors than has previously been reported, the use of negative (psychological distress) and positive (benefit-finding and health-promotion behaviors) responses as independent outcomes, and models adjusted for a wide range of potential confounding variables.
Implications for Nursing
Nurses are uniquely positioned to support unmet needs in MPC survivors. Nurses should be aware that survivorship needs may differ in cancer survivors based upon number of previous cancer diagnoses and that the survivorship experience may differ between MPC and single cancer survivors. In addition, MPC survivors are at an increased risk for psychological distress and may have additional needs related to living with cancer as a chronic illness (e.g., engaging in positive self-management behaviors like healthy diet and exercise). Targeted and ongoing screening for distress in MPC survivors is warranted in specialty and/or primary care settings and may promote early identification and treatment to reduce potential negative downstream health effects.
Oncology nurse scientists should contribute to building the science in this area to identify, understand, and address the unique needs of MPC survivors. As the number of cancer survivors diagnosed with MPC grows, the number of primary cancer diagnoses should be considered in study designs. Although an early body of literature has begun to describe the prevalence of health outcomes in MPC survivors, a paucity of research exists surrounding mechanisms and risk factors for late and long-term effects of cancer and their potentially unique needs. Also unclear is whether the potential for care silos and lack of a clinical home influences health outcomes in MPC survivors. Nurses are well suited to study, assess, and address MPC care needs.

Conclusion
Cancer survivors are increasingly being diagnosed with additional subsequent primary cancers. The current findings provide additional evidence that MPC survivors differ from their single cancer counterparts and are at increased risk for psychological distress. These findings support a need to specifically identify, understand, and address the ongoing, unique needs of MPC survivors. Additional research is needed to identify MPC survivors most at risk for poor outcomes and to understand the care needs and mechanisms that contribute to poor health outcomes in this growing cancer survivor population.
About the Author(s)
Belcher is a doctoral candidate and Robert Wood Johnson Foundation Future of Nursing Scholar in the School of Nursing; Low is an assistant professor in the Department of Medicine and Department of Psychology; and Posluszny is an assistant professor in the Department of Medicine, all at the University of Pittsburgh in Pennsylvania; Schear is the director of mission delivery for the Livestrong Foundation in Austin, TX; Kramer is a nurse resident at the Children’s Hospital of Philadelphia in Pennsylvania; and Donovan is a professor in the School of Nursing and the School of Medicine and vice chair for research in the School of Nursing at the University of Pittsburgh. This research was funded, in part, by a Newmeyer-Thompson Doctoral Student Research Award from the University of Pittsburgh School of Nursing, a Future of Nursing Scholars Award from the Robert Wood Johnson Foundation, grants (K32CA149082, P30CA047904) from the National Cancer Institute, and the Livestrong Foundation (source data). Belcher, Low, Posluszny, and Donovan contributed to the conceptualization and design and provided statistical support. Belcher, Low, and Kramer completed the data collection and provided the analysis. Belcher, Low, Posluszny, Schear, and Donovan contributed to the manuscript preparation. Belcher can be reached at smb208@pitt.edu, with copy to editor at ONFEditor@ons.org. Submitted February 2017. Accepted for publication April 17, 2017.
References
American Cancer Society. (2009). Special section: Multiple primary cancers. In Cancer facts and figures, 2009 (pp. 24–41). Retrieved from https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and…
American Cancer Society. (2014). What are second cancers? Retrieved from http://bit.ly/2frkkIQ
American Cancer Society. (2016). Cancer treatment and survivorship facts and figures, 2016–2017. Retrieved from https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and…
Andrykowski, M.A. (2012). Physical and mental health status of survivors of multiple cancer diagnoses: Findings from the National Health Interview Survey. Cancer, 118, 3645–3653. https://doi.org/10.1002/cncr.26678
Barlow, J., Wright, C., Sheasby, J., Turner, A., & Hainsworth, J. (2002). Self-management approaches for people with chronic conditions: A review. Patient Education and Counseling, 48, 177–187. https://doi.org/10.1016/S0738-3991(02)00032-0
Beckjord, E.B., Reynolds, K.A., van Londen, G.J., Burns, R., Singh, R., Arvey, S.R., . . . Rechis, R. (2014). Population-level trends in posttreatment cancer survivors’ concerns and associated receipt of care: Results from the 2006 and 2010 Livestrong surveys. Journal of Psychosocial Oncology, 32, 125–151. https://doi.org/10.1080/07347332.2013.874004
Begg, C.B. (1999). Methodological and statistical considerations in the study of multiple primary cancers. In A.I. Neugut, A.T. Meadows, & E. Robinson (Eds.), Multiple primary cancers (pp. 13–26). Philadelphia, PA: Lippincott Williams and Wilkins.
Belcher, S., Arida, J., Campbell, G., Hagan, T., Skrovanek, E., & Donovan, H. S. (2015). Exploring well-being, social support, and financial vulnerability in women with recurrent ovarian cancer who report more than one primary cancer diagnosis within the past 3 years [Abstract]. Nursing Research, 64(2), E68. https://doi.org/10.1097/NNR.0000000000000089
Belcher, S.M., Hausmann, E.A., Cohen, S.M., Donovan, H.S., & Schlenk, E.A. (2016). Examining the relationship between multiple primary cancers and psychological distress: A review of current literature. Psycho-Oncology. Advance online publication. https://doi.org/10.1002/pon.4299
Blanchard, C.M., Courneya, K.S., & Stein, K. (2008). Cancer survivors’ adherence to lifestyle behavior recommendations and associations with health-related quality of life: Results from the American Cancer Society’s SCS-II. Journal of Clinical Oncology, 26, 2198–2204. https://doi.org/10.1200/JCO.2007.14.6217
Bluethmann, S.M., Mariotto, A.B., & Rowland, J.H. (2016). Anticipating the “Silver Tsunami”: Prevalence trajectories and comorbidity burden among older cancer survivors in the United States. Cancer Epidemiology Biomarkers and Prevention, 25, 1029–1036. https://doi.org/10.1158/1055-9965.EPI-16-0133
Burris, J.L., & Andrykowski, M.A. (2011). Physical and mental health status and health behaviors of survivors of multiple cancers: A national, population-based study. Annals of Behavioral Medicine, 42, 304–312. https://doi.org/10.1007/s12160-011-9290-0
Campbell, M.K., Tessaro, I., Gellin, M., Valle, C.G., Golden, S., Kaye, L., . . . Miller, K. (2011). Adult cancer survivorship care: Experiences from the Livestrong centers of excellence network. Journal of Cancer Survivorship, 5, 271–282. https://doi.org/10.1007/s11764-011-0180-z
Corbin, J.M., & Strauss, A. (1988). The unending work and care associated with chronic illness. In Unending work and care: managing chronic illness at home (pp. 318–336). San Francisco, CA: Jossey-Bass.
Davies, N., Batehup, L., & Thomas, R. (2011). The role of diet and physical activity in breast, colorectal, and prostate cancer survivorship: A review of the literature. British Journal of Cancer, 105(10), S52–S73. https://doi.org/10.1038/bjc.2011.423
De Gonzalez, A.B., Curtis, R.E., Kry, S.F., Gilbert, E., Lamart, S., Berg, C.D., . . . Ron, E. (2011). Proportion of second cancers attributable to radiotherapy treatment in adults: A cohort study in the US SEER cancer registries. Lancet Oncology, 12, 353–360. https://doi.org/10.1016/S1470-2045(11)70061-4
Dowling, E.C., Chawla, N., Forsythe, L.P., de Moor, J., McNeel, T., Rozjabek, H.M., . . . Yabroff, K.R. (2013). Lost productivity and burden of illness in cancer survivors with and without other chronic conditions. Cancer, 119, 3393–3401. http://doi.org/10.1002/cncr.28214
Dowrick, C., Dixon-Woods, M., Holman, H., & Weinman, J. (2005). What is chronic illness? Chronic Illness, 1, 1–6. https://doi.org/10.1177/17423953050010010901
Ford, E.S., Bergmann, M.M., Kröger, J., Schienkiewitz, A., Weikert, C., & Boeing, H. (2009). Healthy living is the best revenge: Findings from the European Prospective Investigation into Cancer and Nutrition-Potsdam study. Archives of Internal Medicine, 169, 1355–1362. https://doi.org/10.1001/archinternmed.2009.237
Gotay, C.C., Ransom, S., & Pagano, I.S. (2007). Quality of life in survivors of multiple primary cancers compared with cancer survivor controls. Cancer, 110, 2101–2109. https://doi.org/10.1002/cncr.23005
Grady, P.A., & Gough, L.L. (2014). Self-management: A comprehensive approach to management of chronic conditions. American Journal of Public Health, 104(8), e25–e31. https://doi.org/10.2105/AJPH.2014.302041
Harper, F.W.K., Schmidt, J.E., Beacham, A.O., Salsman, J M., Averill, A.J., Graves, K.D., & Andrykowski, M.A. (2007). The role of social cognitive processing theory and optimism in positive psychosocial and physical behavior change after cancer diagnosis and treatment. Psycho-Oncology, 16, 79–91. https://doi.org/10.1002/pon.1068
Hawkins, N.A., Smith, T., Zhao, L., Rodriguez, J., Berkowitz, Z., & Stein, K.D. (2010). Health-related behavior change after cancer: Results of the American Cancer Society’s studies of cancer survivors (SCS). Journal of Cancer Survivorship, 4, 20–32. https://doi.org/10.1007/s11764-009-0104-3
Holland, J.C., Andersen, B., Breibart, W.S., Buchmann, L.O., Compas, B., Deshields, T.L., . . . Freedman-Cass, D.A. (2013). Distress management. Journal of the National Comprehensive Cancer Network, 11, 190–209.
Institute of Medicine. (2006). From cancer patient to cancer survivor: Lost in transition. Washington, DC: National Academies Press.
Kanera, I.M., Bolman, C.A., Mesters, I., Willems, R.A., Beaulen, A.A., & Lechner, L. (2016). Prevalence and correlates of healthy lifestyle behaviors among early cancer survivors. BMC Cancer, 16, 4. https://doi.org/10.1186/s12885-015-2019-x
Klein, W.M., Bloch, M., Hesse, B.W., McDonald, P.G., Nebeling, L., O’Connell, M.E., . . . Tesauro, G. (2014). Behavioral research in cancer prevention and control: A look to the future. American Journal of Preventive Medicine, 46, 303–311. https://doi.org/10.1016/j.amepre.2013.10.004
Knobf, M.T., Cooley, M.E., Duffy, S., Doorenbos, A., Eaton, L., Given, B., . . . Mallory, G. (2015). The 2014–2018 Oncology Nursing Society research agenda. Oncology Nursing Forum, 42, 450–465. https://doi.org/10.1188/15.ONF.450-465
Low, C.A., Beckjord, E., Bovbjerg, D.H., Dew, M.A., Posluszny, D.M., Schmidt, J.E., . . . Rechis, R. (2014). Correlates of positive health behaviors in cancer survivors: Results from the 2010 Livestrong survey. Journal of Psychosocial Oncology, 32, 678–695. https://doi.org/10.1080/07347332.2014.955243
Mariotto, A.B., Rowland, J.H., Ries, L.A., Scoppa, S., & Feuer, E.J. (2007). Multiple cancer prevalence: A growing challenge in long-term survivorship. Cancer Epidemiology, Biomarkers and Prevention, 16, 566–571. https://doi.org/10.1158/1055-9965.EPI-06-0782
Miller, G.E., Cohen, S., & Ritchey, A.K. (2002). Chronic psychological stress and the regulation of pro-inflammatory cytokines: A glucocorticoid-resistance model. Health Psychology, 21, 531–541. https://doi.org/10.1037//0278-6133.21.6.531
Miller, K.D., Siegel, R.L., Lin, C.C., Mariotto, A.B., Kramer, J.L., Rowland, J.H., . . . Jemal, A. (2016). Cancer treatment and survivorship statistics, 2016. CA: A Cancer Journal for Clinicians, 66(4), 271–289. https://doi.org/10.3322/caac.21349
Mitchell, A.J., Chan, M., Bhatti, H., Halton, M., Grassi, L., Johansen, C., & Meader, N. (2011). Prevalence of depression, anxiety, and adjustment disorder in oncological, haematological, and palliative-care settings: A meta-analysis of 94 interview-based studies. Lancet Oncology, 12, 160–174. https://doi.org/10.1016/S1470-2045(11)70002-X
Morton, L.M., Onel, K., Curtis, R.E., Hungate, E.A., & Armstrong, G.T. (2014). The rising incidence of second cancers: Patterns of occurrence and identification of risk ractors for children and adults. In D.S. Dizon (Ed.), 2014 American Society of Clinical Oncology Educational Book (pp. e57–e67). Alexandria, VA: American Society of Clinical Oncology.
Mowls, D.S., Brame, L.S., Martinez, S.A., & Beebe, L.A. (2016). Lifestyle behaviors among US cancer survivors. Journal of Cancer Survivorship, 10, 692–698. https://doi.org/10.1007/s11764-016-0515-x
Mullan, F. (1985). Seasons of survival: Reflections of a physician with cancer. New England Journal of Medicine, 313, 270–273. https://doi.org/10.1056/NEJM198507253130421
Mullan, F. (2016, January). Welcome and keynote lecture: The history of cancer survivorship. In 2016 Cancer Survivorship Symposium. Symposium conducted at the meeting of the American Society Society of Clinical Oncology, San Francisco, CA.
National Cancer Institute. (2017). Metastatic cancer. Retrieved from http://bit.ly/2wWaiFJ
National Comprehensive Cancer Network. (2017a). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Distress management [v.1.2017]. Retrieved from https://www.nccn.org/professionals/physician_gls/pdf/distress.pdf
National Comprehensive Cancer Network. (2017b). NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®): Survivorship [v.1.2017]. Retrieved from https://www.nccn.org/professionals/physician_gls/pdf/survivorship.pdf
Ness, K.K., Wall, M.M., Oakes, J.M., Robison, L.L., & Gurney, J.G. (2006). Physical performance limitations and participation restrictions among cancer survivors: A population-based study. Annals of Epidemiology, 16, 197–205. https://doi.org/10.1016/j.annepidem.2005.01.009
Posluszny, D.M., Dew, M.A., Beckjord, E., Bovbjerg, D.H., Schmidt, J.E., Low, C.A., . . . Rechis, R. (2015). Existential challenges experienced by lymphoma survivors: Results from the 2010 Livestrong survey. Journal of Health Psychology, 21, 2357–2366. https://doi.org/10.1177/1359105315576352
Rechis, R., Reynolds, K.A., Beckjord, E.B., Nutt, S., Burns, R.M., & Schaefer, J.S. (2011). “I learned to live with it” is not good enough: Challenges reported by post-treatment cancer survivors in the Livestrong surveys: A Livestrong report 2010. Austin, TX: Livestrong. Retrieved from http://bit.ly/2yGMrMA
Ryan, P. (2009). Integrated theory of health behavior change: Background and intervention development. Clinical Nurse Specialist, 23, 161–170. https://doi.org/10.1097/NUR.0b013e3181a42373
Shapiro, C.L., McCabe, M.S., Syrjala, K.L., Friedman, D., Jacobs, L.A., Ganz, P.A., . . . Marcus, A.C. (2009). The Livestrong survivorship center of excellence network. Journal of Cancer Survivorship, 3, 4–11. https://doi.org/10.1007/s11764-008-0076-8
Stein, K.D., Syrjala, K.L., & Andrykowski, M.A. (2008). Physical and psychological long-term and late effects of cancer. Cancer, 112(Suppl. 11), 2577–2592. https://doi.org/10.1002/cncr.23448
Thong, M.S., Mols, F., Verhoeven, R.H., Liu, L., Andrykowski, M.A., Roukema, J.A., & van de Poll-Franse, L.V. (2013). Multiple primary cancer survivors have poorer health status and well-being than single primary cancer survivors: A study from the population-based PROFILES registry. Psycho-Oncology, 22, 1834–1842. https://doi.org/10.1002/pon.3227
Underwood, J.M., Townsend, J.S., Stewart, S.L., Buchannan, N., Ekwueme, D.U., Hawkins, N.A, . . . Fairley, T.L. (2012). Surveillance of demographic characteristics and health behaviors among adult cancer survivors—Behavioral Risk Factor Surveillance System, United States, 2009. Morbidity and Mortality Weekly Report Surveillance Summaries, 61, 1–23.

