Myelosuppression, Bone Disease, and Acute Renal Failure: Evidence-Based Recommendations for Oncologic Emergencies
Background: Oncologic emergencies associated with multiple myeloma include myelosuppression (anemia, neutropenia, and thrombocytopenia), bone-related emergencies, and acute renal failure.
Objectives: This article reviews the pathophysiology of these multiple myeloma-associated oncology emergencies and provides a framework for assessment and effective intervention.
Methods: A comprehensive review of the levels of evidence, focusing on assessment, diagnosis, comorbidities, treatment, ongoing monitoring, and patient education, are presented to support the plan of care for at-risk patients.
Findings: Attention to signs and symptoms is the foundation for preventing these emergencies or managing additional escalation of symptoms.
Jump to a section
Symptom management, health maintenance, and survivorship in patients with multiple myeloma (MM) have been areas of focus for the International Myeloma Foundation (IMF) Nurse Leadership Board (NLB). Reviews of myelosuppression, MM bone disease, and renal complications were published in the Clinical Journal of Oncology Nursing in 2008. In the current article, the NLB will again review these important topics and provide evidence-based recommendations, but in the framework of oncologic emergencies (OEs). OEs have been defined as complications resulting from cancer itself, from a paraneoplastic syndrome, or from treatment of the cancer that require prompt interventions to prevent irreversible vital organ damage. OEs can be hematologic, neurologic, or metabolic in origin, but require immediate attention no matter the cause. An understanding of potential OEs, prevention, and prompt intervention is critical to organ health and survival in patients with MM.
The advent of novel therapies has affected the treatment of MM. Initial therapy typically includes a combination of three novel agents, such as an immunomodulatory agent (e.g., lenalidomide or thalidomide), a proteasome inhibitor (e.g., bortezomib or carfilzomib), and a steroid (e.g., dexamethasone or prednisone), followed by an autologous stem cell transplantation in eligible patients (Teh et al., 2014). Following transplantation (or induction therapy for transplantation-ineligible patients), most patients are treated with a reduced-intensity maintenance therapy until progression of disease. Even in patients with relapsed MM, multi-drug combinations are standard, often with the addition of the more recently approved monoclonal antibodies or histone deacetylase inhibitors (Laubach et al., 2016). This treatment paradigm of multi-drug combinations and continuous maintenance therapy has proven to be far more effective than conventional chemotherapy and has significantly improved the overall survival of patients with MM. As a result, there is an ever-increasing population of patients who have received multiple continuous types of treatment with cumulative toxicities, including a decrease in bone marrow cellularity and subsequent myelosuppression. The novel agents for patients with MM have unique side effects and toxicities that are predictable and manageable. When used in combination, however, even mild side effects of individual drugs can be amplified and negatively impact patient quality of life (QOL).
Myelosuppression
The hematologic toxicities of anemia, neutropenia, and thrombocytopenia are a direct result of the various degrees of myelosuppression caused by treatment with immunomodulatory agents, proteosome inhibitors, or monoclonal antibodies. Myelosuppression is defined by the National Cancer Institute ([NCI], 2017) as a condition in which bone marrow activity is decreased, resulting in fewer red blood cells, white blood cells, and platelets. The range of these myelosuppressive hematologic toxicities can vary from minor inconveniences to life-threatening complications. If severe, consequences of under-recognized or untreated myelosupression can lead to OEs, such as life-threatening anemia, neutropenic fever, or bleeding from thrombocytopenia (Miceli, Colson, Gavino, & Lilleby, 2008). Table 1 provides a grading criteria for hematologic toxicity adverse events.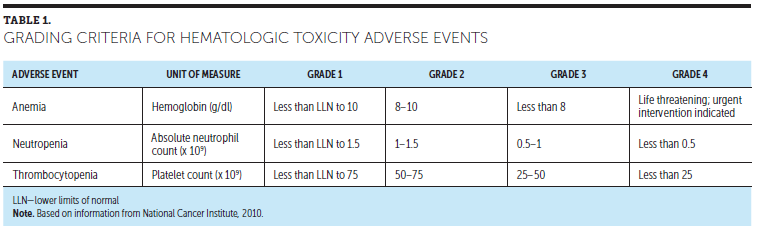
Anemia
Anemia is defined by the NCI (2017) as a condition in which the number of red blood cells is below normal. This results in a reduction in hemoglobin, which decreases the oxygen-carrying capacity of the blood (Miller, 2010). In the grading criteria outlined in the NCI Common Terminology Criteria for Adverse Events (CTCAE), grade 4 anemia is defined as life threatening or disabling and constitutes an OE (National Cancer Institute, 2010). For patients who have been diagnosed with MM, anemia is a common and expected side effect associated with the disease and its treatment. Anemia, even severe, may improve if treatment for MM is successful, but will remain or worsen if the treatment is unsuccessful (Niesvizky & Badros, 2010). Anemia may contribute to inferior outcomes, including reduced survival (Birgegård, GascÓn, & Ludwig, 2006), and patients with uncorrected anemia have a diminished QOL. Therefore, understanding each patient’s risk for developing severe, disabling, or life-threatening anemia is important for its prevention and management.
About 97% of patients with MM develop anemia during the disease trajectory. More than 70% of patients present with anemia at diagnosis, with 8% classified as having severe anemia (Kyle, 1975; Kyle et al., 2003; Maes et al., 2010; Rajkumar & Kumar, 2016). In the European Cancer Anemia Survey, of the 720 patients with persistent or recurrent MM, 85% were anemic at the time of the enrollment. Of these, about 5% had severe anemia, with a hemoglobin less than 8 g/dl (Birgegård et al., 2006). The risk of becoming anemic is highest among patients with MM who initially present with anemia, have persistent or recurrent disease, are female, are older than age 60 years, or are undergoing treatment with platinum chemotherapy (Birgegård et al., 2006). In a study published by Dammacco et al. (1998), 45% of patients with MM receiving chemotherapy required a red blood cell (RBC) transfusion during their treatment.
RBCs, or erythrocytes, are produced in the bone marrow of certain bones (vertebrae, sternum, ribs, pelvis, and proximal femurs), and their production is controlled by the oxygen needs of tissues. Anemia can bring about hypoxia, which in turn triggers the body to begin the process of producing new RBCs. In response to hypoxia, the kidneys release erythropoietin, which sends a signal to erythroid progenitor cells to produce more RBCs. The role of mature RBCs is to serve as a vehicle for hemoglobin transport of oxygen to the tissues and to transport carbon dioxide from the tissues to the lungs. The resulting increase in the oxygen-carrying capacity of the blood functions to downregulate further erythropoietin production (Miller, 2010). Anemia can result from a loss of RBCs from bleeding, from increased RBC destruction, from decreased RBC production, or from a combination of these etiologies. Table 2 provides a review of the etiology of anemia and its contributing factors.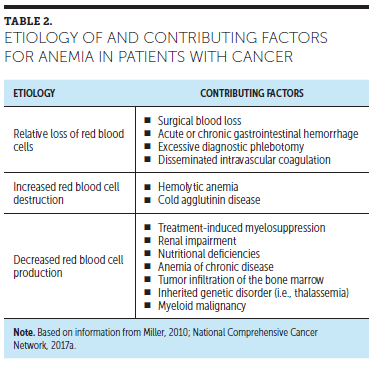
Anemia in MM is caused by many factors, including replacement of bone marrow by plasma cells, which reduces erythroid precursors, leads to erythropoietin deficiency, and makes erythroblast precursors less responsive to erythropoietin; myeloma treatment (both chemotherapy and radiation); and other factors independent of myeloma or treatment itself (Niesvizky & Badros, 2010). The major cause of anemia in patients with MM is myeloma cells inducing the death of erythroblasts (RBC precursors) (Terpos et al., 2015). Cancer cells also produce signaling molecules (cytokines) that result in the sequestration of iron away from RBCs, which decreases RBC production and may shorten RBC survival (National Comprehensive Cancer Network [NCCN], 2017a). Bone marrow infiltration by plasma cells also reduces the production of hepcidin, which impairs the use of iron and increases plasma volume via paraproteins (Maes et al., 2010; Terpos et al., 2015).
Signs and Symptoms
Recognizing the signs, symptoms, and risks for severe anemia can mitigate potentially life-threatening anemia. Life-threatening anemia often is a preventable OE. Table 3 lists tests to establish anemia in patients with MM. Signs and symptoms of all grades of anemia severity may include pallor of the skin and mucous membranes, shortness of breath, heart palpitations, soft systolic murmurs, lethargy, and fatigue (NCI, 2010). Fatigue, which is the most common side effect of anemia, can reduce QOL by limiting the ability to work and otherwise influence physical and emotional well-being (Miceli et al., 2008). The symptoms of anemia are likely to be pronounced if the anemia presents acutely; however, if it develops gradually, the body can compensate for the lower oxygen-carrying capacity of the blood by increasing cardiac output and coronary flow, altering blood viscosity, and adjusting oxygen consumption and extraction. The presence of preexisting cardiovascular, pulmonary, or cerebral vascular disease may interfere with these compensatory mechanisms, making the patient less able to tolerate even mild anemia and can lead to an emergency situation (NCCN, 2017a).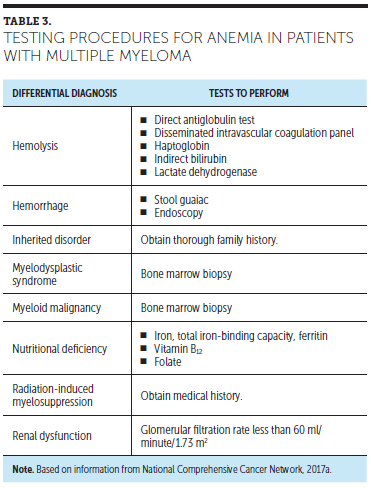
Neutropenia
Neutropenia is defined by the NCI (2017) as a condition in which there is a lower-than-normal number of neutrophils (a type of white blood cell). All grades of neutropenia severity can lead to OEs, such as neutropenic fever, pneumonia, and sepsis in patients who already harbor functional immunoglobulin deficiency (Miceli et al., 2008). In patients with MM, neutropenia may be caused by the disease or by its treatment. When present in high numbers, plasma cells directly interfere with normal hematopoiesis and neutrophil production. This disease-related neutropenia is common in patients at diagnosis and late in the disease (Teh et al., 2014). Beyond neutropenia, disease-related immunodeficiency also involves B-cell dysfunction; hypogammaglobulinemia; and T-cell, NK-cell, and dendritic-cell abnormalities (Teh et al., 2014). Because MM is a disease afflicting older adults, many patients have both functional and physiologic age-related comorbidities that predispose them to increased risks of infection. Given this combination of factors, patients with MM are at significantly greater risk for developing bacterial, viral, and fungal infections than matched controls, and infections are a leading contributor to morbidity and mortality. The most frequent bacterial infections (listed in order of prevalence) are pneumonia, septicemia, cellulitis, and pyelonephritis. Herpes zoster and influenza are the two most common viral infections (Blimark et al., 2015). As the disease progresses and immune dysfunction worsens, the spectrum of causative agents widens (Teh et al., 2015). Therefore, even mild neutropenia can represent an OE in patients with MM due to many factors.
Traditional chemotherapy agents, such as melphalan or cyclophosphamide, greatly increase the risk of infection through a combination of rapid, prolonged neutropenia and disruption of physical defense barriers, such as the mucosa. Such chemotherapy agents are not widely used for the treatment of MM except during an autologous stem cell transplantation and for disease control late in the disease trajectory. Although newer treatment agents (immunomodulatory drugs and proteasome inhibitors) also negatively affect neutrophil counts, they have other effects on the immune system, some of which are beneficial. In fact, in a longitudinal study, neither class of these agents was independently associated with an increased risk of infection (Teh et al., 2015). However, these newer agents are rarely used as monotherapies and are most often combined with each other and with corticosteroids. Corticosteroids are highly immunosuppressive even when used as single agents and significantly increase the risk of developing infection (Teh et al., 2014). When corticosteroids are combined with lenalidomide or pomalidomide, neutropenia rates have been reported to be as high as 39% and 50%, respectively (Dimopoulos et al., 2014; Dimopoulos, Palumbo, et al., 2016). Therefore, in the era of combination therapy, the risk of severe neutropenia and subsequent fever has increased (Teh et al., 2014).
Signs and Symptoms
Like other hematologic toxicities, neutropenia is graded according to NCI CTCAE criteria. Neutropenia itself is not an OE, but the risk of developing an infection is directly related to the depth and duration of the neutropenic episode. Bodey, Buckley, Sathe, and Freireich (1966) reported that the most important risk factor for developing an infection was an absolute neutrophil count of less than 0.5 x 109. Because adequate neutrophils are necessary to mount an inflammatory reaction at the site of an active infection, fever may be the first and only sign of a bacterial infection in profoundly neutropenic patients. Fever is defined as a single oral temperature of 38.3°C (101°F) or higher, or two separate temperatures of 38°C (100.4°F) or higher, measured one hour apart (Hughes et al., 2002). Local symptoms, such as erythema, induration, or purulent discharge, may not be present in the absence of pus-generating neutrophils and monocytes. Likewise, pulmonary infiltrates are often absent in patients who are neutropenic because of a diminished and delayed inflammatory response (Lewis, Hendrickson, & Moynihan, 2011). In the absence of fever, other symptoms associated with a local or systemic infection may include chills, rigors, cough, dyspnea, nausea, diarrhea, dysuria, dizziness, fatigue, and pain. Therefore, when assessing a patient with neutropenia, a thorough review of systems is critical. In addition, a careful physical examination should focus on areas of pain and should include a careful inspection of the oral mucosa, skin, and any venous access devices.
Thrombocytopenia
Thrombocytopenia is defined by the NCI (2017) as a condition in which there is a lower-than-normal number of platelets in the blood and is the primary cause of bleeding in patients with all types of cancer. When platelet levels fall below normal levels, adult patients are considered thrombocytopenic, and their thrombocytopenia can be further subdivided into mild, moderate, and severe subgroups as per NCI (2010) CTCAE criteria. Although platelet count is the best indicator of the potential risk of bleeding, the correlation is imprecise and varies based on the underlying condition and other clinical factors (Avvisati, Tirindelli, & Annibali, 2003). Severe thrombocytopenia is considered a life-threatening OE. Because of this, the number of platelets considered “safe” lacks well-defined evidence, and there is no established threshold above which platelets should be maintained to avoid bleeding. Spontaneous bleeding rarely occurs when platelets are more than 50 x 109/L, and risk of bleeding generally increases only when platelets fall below 20 x 109/L (Avvisati et al., 2003).
In patients with MM, thrombocytopenia often is present at diagnosis and often is most severe at end-stage disease. During that time, the large numbers of MM cells present in the bone marrow play a significant role in the pathogenesis. Thrombocytopenia is also a common toxicity of proteasome inhibitors used for the treatment of MM. However, for these agents, thrombocytopenia is typically cyclical, predictable, and generally reversible. Patients who are being treated with an immunomodulatory agent or carfilzomib should also take antithrombotic agents (i.e., aspirin, enoxaparin) to reduce their risk of thrombotic events related to these drugs. Because this increases the risk of bleeding, discontinuation of anticoagulation or antiplatelet therapy needs to be considered when platelet levels decrease, particularly when platelets fall below 50 x 109/L (Noonan et al., 2017). Steroids, which are an integral part of most anti-myeloma regimens, adversely affect the gastric mucosa, impair wound healing, and lead to an increased risk of bleeding (King & Faiman, 2017). Although onset of treatment-related thrombocytopenia may be gradual, thrombocytopenia may appear rapidly with both viral and bacterial infections related to platelet consumption, or it may be attributed to the drugs used to treat the infection. Some drugs, such as vancomycin, result in frank thrombocytopenia, whereas others increase risk of bleeding related to impaired platelet function (Aster & Bougie, 2007).
Signs and Symptoms
Bleeding caused by thrombocytopenia may manifest in minor signs and symptoms, such as ecchymosis or petechiae, or in more overt symptoms such as epistaxis, hemoptysis, hematuria, hematemesis, melena, vaginal bleeding, or oozing from skin lesions or vascular access lines. Hemorrhage may also occur as a catastrophic event, such as a major cerebral or gastrointestinal bleed (Damron et al., 2009). As such, nurses should conduct thorough patient histories to assess for both frank and occult bleeding. Although noting the level of thrombocytopenia identified by complete blood count (CBC) is critical to assessing risk of bleeding, noting the hemoglobin level is also of importance because the rapid development of anemia may indicate a possible hemorrhage.
Evidence-Based Recommendations for the Prevention and Management of Myelosuppression
Level of Evidence I
• The prevention and management of severe anemia in MM must include treatment of the underlying malignancy, administration of erythropoiesis-stimulating agents (ESAs) if indicated, correction of additional underlying causes, and transfusions with packed RBCs with or without iron supplement (NCCN, 2017a).
• Serum creatinine must also be checked, as anemia may become severe if renal function decreases (Faiman, Mangan, Spong, & Tariman, 2011).
• ESAs for patients with MM should be used after an assessment of the benefits and risks. Benefits include a potential decrease in the number of RBC transfusions. Several prospective studies have shown that ESAs can increase hemoglobin levels by 2 g/dl or more in 60%–75% of patients with MM with symptomatic anemia (Terpos et al., 2015). Potential risks of ESAs include an increase in the number of thrombotic events, hypertension, seizures, pure red cell aplasia, and possible decreased survival related to shortened time to tumor progression (NCCN, 2017a).
• Underlying causes of anemia that may not be attributable to MM should be treated. Anemia in cancer may be attributed to underlying comorbidities, such as bleeding, hemolysis, hereditary disease, renal insufficiency, nutritional deficiencies, anemia of chronic disease, or a combination of these factors (NCCN, 2017a). The mean corpuscular volume will define whether a patient has a microcytic (less than 80 fl), macrocytic (greater than 100 fl), or normocytic (80–100 fl) anemia. Microcytic anemia may be related to iron deficiency, thalassemia, anemia of chronic disease, or sideroblastic anemia. Macrocytic anemia may be related to medications, alcoholism, myelodysplastic syndrome, or vitamin B12 or folate deficiencies. Normocytic anemia may be a result of hemorrhage, hemolysis, bone marrow failure, anemia of chronic inflammation, or renal insufficiency (NCCN, 2017a).
• Leukocyte-reduced packed RBC is recommended for asymptomatic patients with MM at high risk for complications from anemia, such as patients who have had a progressive decline in their hemoglobin following intensive therapy or patients with comorbidities such as cardiac disease, chronic pulmonary disease, or cerebral vascular disease (NCCN, 2017a). Leukocyte-reduced blood products decrease the risk of febrile reactions, human leukocyte antigen alloimmunization, and cytomegalovirus infection (Miceli et al., 2008).
• Select patients may refuse blood transfusions because of either religious or personal preferences. Figure 1 outlines preventive management of anemia for these patients. In extreme cases of severe, life-threatening anemia, pure oxygen (400 mmHg, SaO2 = 1) has been used to increase blood oxygenation (NCCN, 2017a). The benefit of an RBC transfusion in the symptomatic patient is a prompt increase in the hemoglobin and a potential for rapid improvement in anemia-related symptoms (NCCN, 2017a) (see Figure 2). 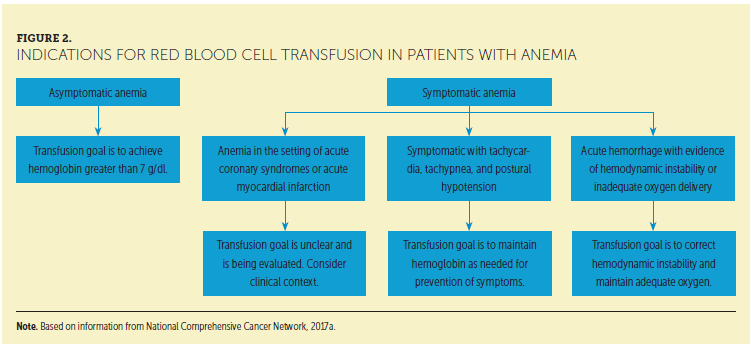
• Antibacterial and antifungal prophylaxis is recommended for patients with MM at high risk for infection, such as older adults, frail patients, or those who have a history of infections or cardiopulmonary comorbidities. Based on clinical guidelines, this same group may also benefit from the prophylactic use of hematopoietic growth factors, such as filgrastim or newer biosimilars (NCCN, 2017a; Smith et al., 2015).
• It is recommended that all patients receive vaccination against influenza, Haemophilus influenza, and Streptococcus pneumoniae, although vaccine efficacy is not guaranteed because patients with MM may not mount a robust immune response. Live vaccines, such as the varicella-zoster virus vaccine, should not be given to patients with MM, as live vaccines have the potential to cause the disease they are meant to prevent (Flowers et al., 2013; Freifeld et al., 2011; NCCN, 2017c; Snowden et al., 2011).
Level of Evidence II
• All patients receiving therapy for MM should have their CBC closely monitored, with specific frequency and timing of monitoring based on the prescribing information for each drug or combination of drugs. Dose modifications or treatment delays should be considered if grade 3 anemia, neutropenia, or thrombocytopenia occurs (Amgen Inc., 2016a; Bristol-Myers Squibb, 2015; Celgene, 2016, 2017a, 2017b; Janssen Biotech, 2017; Millennium Pharmaceuticals, 2017; Novartis, 2016; Takeda Oncology, 2016).
• Patients undergoing treatment with anti-myeloma therapy should observe the hematologic outcomes achieved solely through tumor reduction prior to considering the use of ESAs. If anemia does not improve with anti-myeloma therapy, treatment with ESAs may be considered, beginning with the lowest possible dose (Rizzo et al., 2010; Terpos et al., 2015).
• The use of ESAs during chemotherapy is recommended, particularly in patients with renal impairment, when the hemoglobin concentration is less than 10 g/dl and no improvement is noted in anemia despite MM disease response (Palumbo et al., 2014).
• Caution should be exercised when using ESAs with other chemotherapeutic agents (i.e., immunomodulatory drugs, doxorubicin, or high-dose corticosteroids) that increase the risk of thromboembolic complications (Rizzo et al., 2010). A few randomized studies have evaluated the impact of ESAs on outcomes in patients with MM. One randomized study showed no evidence of inferior outcome following ESA treatment; however, the trial was small and statistically underpowered (Richardson et al., 2011).
• To minimize the risk of infection, the use of neutropenic precautions is recommended in patients with MM (see Figure 3). Patients and their caregivers should be educated to promptly recognize and act on signs and symptoms of infection and should be given specific written instructions on how and when to contact a healthcare provider (Flowers & Karten, 2013).
• Prophylactic antivirals are recommended for patients receiving proteasome inhibitors (Amgen Inc., 2016a; Millennium Pharmaceuticals, 2017; Takeda Oncology, 2016) and following autologous stem cell transplantation.
• For patients receiving high-dose corticosteroids, prophylaxis with trimethoprim-sulfamethoxazole should also be considered (Flowers et al., 2013). The use of prophylactic immunoglobulin replacement is not uniformly recommended, although it may be useful in subsets of patients with hypogammaglobulinemia and recurrent infections.
• All healthcare workers and caregivers should follow hand hygiene guidelines and respiratory and cough etiquette guidelines to reduce pathogen exposure. In addition, healthcare personnel who access central venous catheters should follow well-established guidelines to prevent infection through these devices (NCCN, 2017c; O’Grady et al., 2011; Schiffer et al., 2013).
• For patients who develop fever and neutropenia, urgent action is required because these patients may quickly become hemodynamically unstable. Paired blood cultures should be obtained prior to starting antibiotics, and if clinical signs or symptoms are present, cultures of the nares, urine, or diarrheal stools may be obtained. All patients should receive initial empirical antibiotic therapy within an hour of triage to cover gram-positive and gram-negative pathogens. Although most patients with febrile neutropenia will be admitted to the hospital, a select low-risk group may be managed safely in the outpatient setting (Flowers et al., 2013; Freifeld et al., 2011).
• Platelet transfusion practices should follow established guidelines, such as those provided by the American Society of Clinical Oncologists (Schiffer et al., 2001). Although invasive procedures should be minimized in patients with thrombocytopenia, a platelet count of 40 x 109/L to 50 x 109/L is considered acceptable for proceeding with invasive procedures in the absence of other coagulation abnormalities.
Level of Evidence VII
• Beyond platelet transfusions, patients should avoid using drugs that adversely affect platelet function, such as aspirin, ibuprofen, or naproxen, unless medically necessary and directed by their healthcare provider to take them for thromboprophylaxis (Miceli et al., 2008).
• When platelets are low, patients should avoid activities that may result in bleeding or bruising, such as contact sports and tattooing, and they should be instructed to use soft sponges and nonabrasive toothpaste for their oral care (Miceli et al., 2008).
Bone Disease
About 90% of people will, at some point in the disease continuum, have lytic lesions visible by radiographic imaging. At the time of diagnosis, about 80% of patients will have bone disease and 60% of patients will present with bone pain (Kyle et al., 2003; Terpos, Dimopoulos, & Moulopoulos, 2016). The most common bones affected are the vertebrae, long bones, and flat bones (Roodman, 2008). This high incidence of bone disease leads to an increased risk of two significant OEs in the MM population, hypercalcemia of malignancy (HOM) and spinal cord compression (SCC), both at time of diagnosis and at fulminant relapse.
Hypercalcemia of Malignancy
Calcium is an abundant mineral in the body and a common nutrient in dairy and some green leafy vegetables. Among its many functions, calcium is essential for bone development and maintenance, muscle contractility, and transmission of nerve impulses. Normal calcium levels range from 8.5–10.5 mg/dl, with hypercalcemia being defined as serum levels greater than 10.5 mg/dl. Hypercalcemia can result from hyperparathyroidism, but HOM is related to bone metastasis from a malignant process (Terpos et al., 2015).
HOM is not unique to MM, occurring in about 20% of patients with advanced solid tumors (Kaplan, 2012a; Pace, 2015). However, with the high incidence of bone disease in MM, HOM is a potential OE at time of diagnosis and relapse, and 15%–40% of patients with MM present with HOM (Kaplan, 2012a; Roodman, 2008). In MM, bone disease is closely connected to the bone marrow microenvironment, and an imbalance of osteoclast and osteoblast formation results in decreased bone formation, calcium resorption, and lytic lesions (Galson, Silbermann, & Roodman, 2012).
The greatest concentration of the body’s calcium is held in the bone. The remaining 1% of calcium is found in the serum, with a high concentration primarily bound to albumin (Kaplan, 2012a; Pace, 2015). This is particularly important in MM because many patients will have an abnormally low albumin level with active disease (Greipp et al., 2005). Therefore, when reviewing for hypercalcemia in a patient with MM, albumin must also be taken into consideration. A serum calcium level adjusted for hypoalbuminemia is higher than the reported level (Kaplan, 2012a). About 20% of newly diagnosed patients with MM will present with a calcium level less than 10.5 mg/dl (Faiman & Bilotti, 2014; Pace, 2015; Shane & Berenson, 2015; Thomas & Chung, 2016).
Signs and Symptoms
Treatment of HOM is directed by patient symptoms and serum calcium level. If treated, there is an 80% rate of reversal; if untreated, hypercalcemia can result in renal failure and death (Terpos et al., 2015). Hypercalcemia can be mild (less than 12 mg/dl), moderate (12–14 mg/dl), or severe (greater than 14 mg/dl). Lethargy, constipation, dysuria, polydipsia, confusion, muscle weakness, and cardiac dysrhythmias are symptoms that patients may exhibit as calcium levels rise. Not all patients are symptomatic, or their symptoms may be attributed to the underlying malignancy or treatment (Terpos et al., 2015) (see Figure 4).
Astute nursing assessment and rapid nursing interventions are important when reducing high calcium levels and reversing the signs and symptoms associated with HOM. The primary focus is to reduce serum calcium quickly and safely and then to identify and treat the underlying cause of HOM (see Figure 5). Administration of prescribed calcium-reducing medications in a timely manner is essential. Dialysis may be needed if HOM is associated with acute renal failure. Additional diagnostic testing and disease reassessment should be anticipated, as HOM likely indicates active or relapsed disease. While hospitalized, patients should be monitored closely for risk of falls and wandering related to lethargy and confusion. Fluid volume overload is possible during times of active hydration; intake and output need to be closely monitored, particularly in patients with renal dysfunction and cardiac comorbidities. If patients have not previously received IV bisphosphonate therapy, they should be monitored for first-infusion reaction of fever, chills, and body aches (Miceli, Colson, Faiman, Miller, & Tariman, 2011). If adjunct steroids are administered to a patient with diabetes, glucose levels should be monitored frequently. HOM may be associated with generalized bone and/or fracture pain. Analgesia may be needed (Rome, Noonan, Bertolotti, & Miceli, 2017), but both HOM and opioid analgesics can cause constipation. Patient education should include the caregiver and should be reinforced with the patient when their calcium levels decrease to normal range (Faiman & Bilotti, 2014; Kaplan, 2012a; Pace, 2015; Shane & Berenson, 2015) (see Table 4).
Spinal Cord Compression
Patients with MM are at risk for SCC, another bone disease–related OE. In MM, SCC can be caused by a destructive lesion involving the vertebrae or by an extra-osseous plasmacytoma. In both circumstances, the spinal cord is compromised, resulting in back pain with progression to neurologic deficit if untreated (Ha, Kim, & Kim, 2013; Kaplan, 2012b; Roodman, 2008).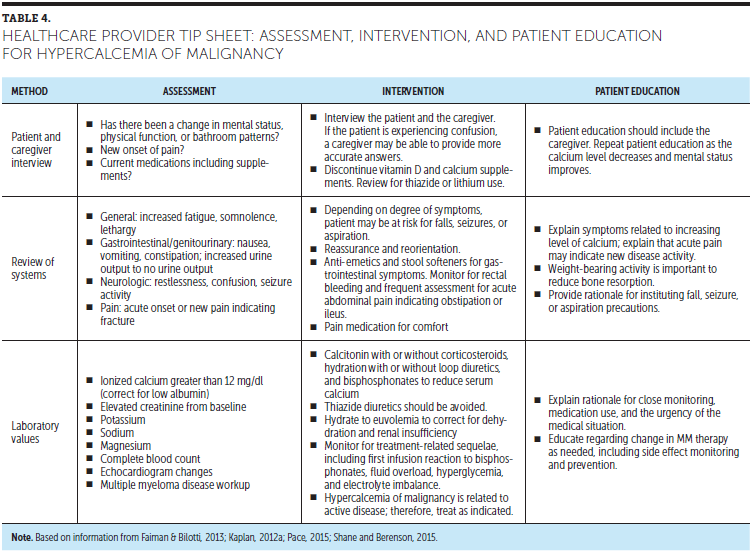
The spine is the part of the skeletal structure that provides stability and movement. It is made up of individual vertebrae identified in sections by their anatomical location—cervical (7), thoracic (12), lumbar (5), and the sacrum and coccyx. The vertebrae act as a skeletal protector of the spinal cord. The spinal cord is part of the central nervous system and has spinal nerve branches. These branches become peripheral nerves and supply motor and sensory fibers to areas of muscle (myotomes) and skin (dermatomes) (Clemente, 1985; Kaplan, 2012b).
As previously discussed, MM bone disease is a frequent occurrence at time of diagnosis (60%–80%) and during the course of the disease (90%) (Kyle et al., 2003; Terpos, Christoulas, et al., 2016). About 50% of people with bone disease will have spinal involvement (Roodman, 2008). Spinal involvement occurs on a spectrum from focal lesions to osteolytic lesions to osteoporosis, any of which can lead to vertebral compression fracture (Hussein et al., 2008). An estimated 30% of patients with spinal MM bone disease will experience vertebral compression fracture, whereas the incidence of SCC is less than 5% (Dispenzieri & Kyle, 2005; Hussein et al., 2008; Roodman, 2008). However, MM accounts for 11% of all neoplastic incidence of SCC complications (Terpos et al., 2015). SCC is more commonly a result of an extradural destructive vertebral lesion, or bone plasmacytoma, and less commonly related to an extra-osseous (or soft tissue) plasmacytoma, which is seen more often later in the disease (Dispenzieri & Kyle, 2005; Ha et al., 2013).
Signs and Symptoms
Presenting symptoms of SCC include back pain and neurologic symptoms that depend on the level and extent of the cord compression (Ha et al., 2013; Kaplan, 2012b; Roodman, 2008). Pain onset may be acute and followed by progression of neurologic deficits to paralysis in hours. The thoracic region is the most common area of spinal cord involvement, with symptoms experienced distal to the area of involvement (Dispenzieri & Kyle, 2005). Therefore, neurologic symptoms may include weakness and paresthesia extending from the abdomen to the lower extremities; bowel, bladder, and sexual dysfunction; and paralysis (Kaplan, 2012b). Symptoms that may indicate a higher level of spinal involvement should not be ignored and may include upper body weakness and/or paresthesia, respiratory insufficiency, and the Lhermitte sign or Horner Syndrome (Kaplan, 2012b).
If a patient presents with symptoms concerning for SCC, a thorough neurologic evaluation should be performed. Interview the patient regarding personal history (cancer, previous vertebral compression fracture); symptoms, particularly pain and neurologic changes (onset, duration, intensity); and any interventions already attempted (pain medication, physical therapy). Comorbid or preexisting conditions need to be considered when interviewing the patient. For example, urinary incontinence, paresthesia, and neuropathy may be preexisting and not related to current symptoms. Physical examinations should include spinal palpation and a full neurologic assessment (Kaplan, 2012b).
Diagnostic evaluation may start with plain radiographs because of their availability and cost; however, they are less reliable for evaluating SCC than other diagnostic imaging modalities. Imaging techniques have become more sensitive and, therefore, the preferred methods to assess MM bone disease have evolved. Without controversy, the preferred imaging for the spine is magnetic resonance imaging (MRI) (Dimopoulos et al., 2015; Terpos, Christoulas, et al., 2016). Spinal MRI should be performed to evaluate for SCC because it clearly defines the area of compression, which will aid in developing a plan for intervention (see Table 5).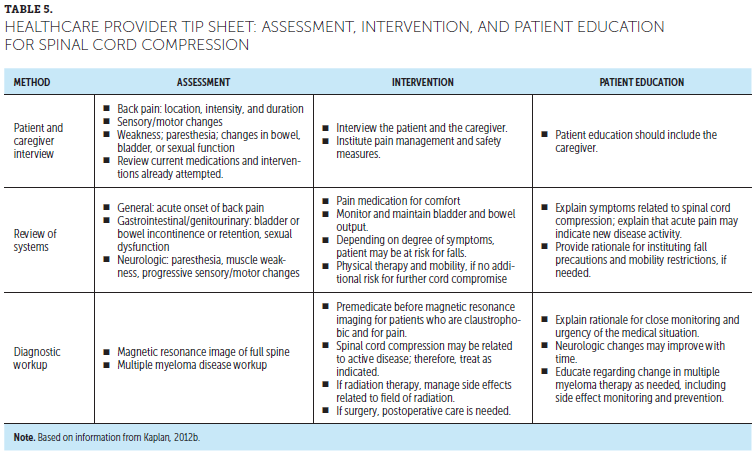
SCC is an OE requiring immediate intervention to prevent progression of symptoms and nerve damage. Educate patients on signs and symptoms of SCC and the need to contact a healthcare provider if back pain occurs with acute onset. For a diagnosis of SCC, nursing assessment is needed for pain, safety, and sensory and motor function. Interventions are focused on safety and pain management, and based on a plan of care. If radiation is prescribed, manage side effects based on the field of radiation.
Evidence-Based Recommendations for the Management of Bone-Related Oncologic Emergencies
Level of Evidence I
• Treating HOM concurrently with treatment of its underlying cause, such as active myeloma, is recommended (Faiman & Bilotti, 2014; Kaplan, 2012a; NCCN, 2017b; Pace, 2015; Shane & Berenson, 2015; Thomas & Chung, 2016).
• Local external-beam radiation should be used at a palliative dose of 10–30 Gy as part of frontline treatment for SCC, particularly when neurologic symptoms are present. A therapeutic dose of 40–50 Gy may be used, and is potentially curative, if the cause of SCC is a solitary plasmacytoma, which is rare (Dispenzieri & Kyle, 2005; Hussein et al., 2008; NCCN, 2016; Terpos, Morgan, et al., 2013).
Level of Evidence II
• For symptomatic and severe hypercalcemia, immediate intervention is recommended, which likely involves hospitalization. Calcitonin and saline hydration can reduce serum calcium levels within 48 hours. Adjunct use of bisphosphonate therapy (IV zoledronic acid or pamidronate) as part of frontline therapy becomes effective in days, providing later calcium control. Mild to moderate hypercalcemia that is asymptomatic is not an OE, but does require intervention. Patients should be instructed on how to administer oral hydration and be educated about weight-bearing activity. In addition, patient medications that may contribute to higher levels of blood calcium should be identified, including lithium, vitamin D, and calcium supplements, (Kaplan, 2012a; Faiman & Bilotti, 2014; Pace, 2015; Shane & Berenson, 2015; Thomas & Chung, 2016).
• Denosumab, a monoclonal antibody against eceptor activator of nuclear factor kappa-B ligand, can be used for patients not responding to bisphosphonate treatment and is not restricted by renal function (Shane & Berenson, 2015; Thomas & Chung, 2016)
• Calcitonin has a rapid but short-term effect with onset of action within four to six hours. Corticosteroids such as hydrocortisone or prednisone can be added to prolong the effectiveness of calcitonin, but neither corticosteroids nor calcitonin should not be used as single agents (Sternlicht & Glezerman, 2015).
• The goal of hydration is currently to achieve euvolemia by replacing volume loss from nausea and renal dysfunction that can contribute to hypercalcemia. Aggressive hydration with concomitant use of loop diuretics (i.e., furosemide) is less favored because of the availability of bisphosphonate agents (Shane & Berenson, 2015) and the desire to avoid complications that can arise from electrolyte imbalance and comorbid conditions.
• Regardless of the cause, SCC should be considered an OE that requires immediate intervention to prevent irreversible nerve damage. First-line therapy includes a high-dose steroid infusion, and although no consensus exists on dose and type, use of dexamethasone, as much as 100 mg IV bolus, followed by a tapering oral dose for days to weeks, is reported in the literature (Dispenzieri & Kyle, 2005; Kaplan, 2012b).
• MM tends to be very radiosensitive, and external beam radiation results in improvement in motor function in 75% of patients. Surgical intervention is less common but may be warranted, particularly if vertebral bone fragments compromise the spinal cord, or plasmacytoma is resistant to radiation. An orthopedic specialist should be consulted (Terpos, Morgan, et al., 2013). The use of spinal augmentation with vertebroplasty or kyphoplasty is contraindicated in spinal cord compression (Hussein et al., 2008).
Acute Renal Failure
The kidneys are vital organs, responsible for flushing out toxins and ridding the body of waste products. When the kidneys are unable to function properly because of acute or chronic insults, patients may be forced to begin dialysis, an artificial method of waste product removal. In addition, if acute renal failure and acute kidney injury occur, the individual may be unable to receive future anti-myeloma therapies. Severe and permanent renal failure can follow acute renal failure, making treatment decisions more difficult and affecting overall survival for patients with MM. Chronic or acute insults to the kidney can place patients at risk for end-stage renal disease, which requires acute or chronic dialysis. However, prompt diagnosis and reversal of renal toxicity can lead to renal recovery, and the poor prognostic significance of acute kidney injury for future MM treatment options can lessen over time (Doshi, Lahoti, Danesh, Batuman, & Sanders, 2016; Hsu & Hsu, 2016).
Therefore, it is well within the scope of nurses to participate in the routine assessment and monitoring of kidney function in patients with MM. With education focused on primary prevention and prompt identification of acute kidney injury and appropriate nursing management, renal function can be recovered. In contrast, a lack of attention to these measures can lead to grave outcomes for patients (Hsu & Hsu, 2016).
Definition and Etiology of Acute Renal Failure
As a potential OE, acute renal failure is defined as an acute elevation in serum creatinine levels, which is accompanied by a decrease in glomerular filtration rate (GFR) levels within a short time period (Dimopoulos, Sonneveld, et al., 2016). Many criteria exist with regards to acute renal failure severity; however, any rapid decline in renal function can be considered an OE, particularly among patients with active MM (Terpos et al., 2015). Identifying the underlying cause of renal injury and employing prompt and appropriate interventions to address the renal failure are crucial steps towards reversing the injury and preventing further damage.
The etiology of acute renal failure in MM includes active infections, dehydration, the use of nephrotoxic medications, as well as comorbidities such as uncontrolled diabetes and hypertension. In MM, acute tubular necrosis is a common cause of acute renal failure that may be caused by dehydration in the presence of urinary light chains, which overwhelm the distal tubules. Diuretic medications, such as furosemide, can lead to urinary cast formation, increased serum creatinine levels, and possibly acute kidney injury. Hypercalcemia, a common symptom of MM when the disease is active, can lead to vasoconstriction (Terpos, Christoulas, et al., 2013). Nonsteroidal anti-inflammatory drugs (NSAIDs) and aminoglycoside antibiotic use can also decrease blood flow to and from the kidneys (Faiman et al., 2011). Given these risk factors for renal failure, active myeloma remains a common cause of renal failure. The pathogenesis of acute renal failure and cast nephropathy, which is the most common type of renal injury in MM, is reviewed in Faiman et al. (2017).
When disease-related acute renal failure occurs, it is important to promote hyper-hydration, with close attention to the patient’s risk of fluid volume overload (Faiman et al., 2011). Rapid control of the disease is necessary to give the kidney the best chance at returning to the baseline function and preventing additional impairment. As such, prompt treatment of active MM disease with corticosteroids, bortezomib, and other effective treatment strategies can assist with renal recovery (Dimopolous, Sonneveld, et al., 2016).
Signs and Symptoms
Patients should be counseled to avoid nephrotoxic agents, such as over-the-counter pain medications like NSAIDs, which can decrease renal blood flow and, in the presence of immunoglobulin light chains, obstruct the tubules of the kidneys, causing a decrease in creatinine clearance and severe renal failure (Faiman et al., 2011).
Adequate oral hydration is also very important for patients. Dehydration can happen quickly and result in acute kidney injury, which can lead to permanent kidney failure if uncorrected. Assessing the patient’s oral intake at each appointment is necessary in helping to prevent dehydration from occurring. Encouraging patients to drink plenty of hydrating fluids and to avoid excessive amounts of caffeinated beverages are key tools in protecting the kidney from dehydration and/or injury. Treatment of the underlying cause of renal failure with effective anti-myeloma therapies may be needed. Common drugs to treat MM and their considerations in regards to dose reduction is located in Fairman et al. (2017) in this supplement.
Evidence-Based Recommendations for the Prevention and Management of Acute Kidney Injury
Level of Evidence I
• Identify and correct the causes of acute kidney injury, including dehydration, hypovolemia, hypotension, loop diuretics, angiotensin-converting enzyme inhibitors, angiotensin II receptor inhibitors, NSAIDs, aminoglycoside antibiotics, hypercalcemia, infection, sepsis, or iodinated contrast dye. This must be a top priority action for the nurse clinician to prevent long-term and permanent renal complications. In many cases, adequate IV or oral hydration (with caution to avoid overhydration) can help to reverse acute kidney injury or acute renal failure related to dehydration, hypovolemia, hypotension, and an acute tubular necrosis-type of kidney injury (Gavriatopoulou, Terpos, Kastritis, & Dimopoulos, 2016; Moos, van Vemde, Stoker, & Bipat, 2013).
• Clinicians should assess patients for reversible and nonmodifiable acute kidney injury risk factors before any use of oral or IV iodinated contrast dye for radiologic studies. The nonmodifiable factors that can increase the risk of acute kidney injury following iodinated contrast dye include chronic kidney disease (CKD), diabetes, heart failure, low body mass index, and aging (Faiman et al., 2011) (see Figure 6).
• A serum creatinine measurement should be obtained through a basic chemistry panel prior to any radiologic procedure that would require an oral or IV iodinated contrast dye. Once a creatinine level is available, an estimated GFR can be calculated by using the Chronic Kidney Disease–Epidemiology (CKD-EPI) equation (National Institute of Diabetes and Digestive and Kidney Diseases [NIDDKD], 2016), which accounts for the patient’s age, creatinine level, gender, and race (Terpos, Christoulas, et al., 2013).
• Contrast-induced nephropathy risk assessment and prophylactic strategies should be based on GFR estimated from the CKD-EPI equation rather than solely relying on the absolute creatinine value. The European Renal Best Practice Ad-Hoc Working Group (Fliser et al., 2012) on contrast-induced nephropathy and a study by Terpos, Christoulas, et al. (2013) demonstrated better detection of stage III–V CKD (stage III with estimated GFR 30–60 ml/minute/173 m2) using the CKD-EPI equation than using the creatinine value alone in newly diagnosed myeloma.
Level of Evidence II
• High-cutoff dialyzers for plasmapheresis are recommended in conjunction with anti-myeloma therapies. Plasmapheresis can effectively, albeit temporarily, remove damaging paraproteins and has been shown to improve or reverse acute renal failure based on data from a few small studies (Burnette, Leung, & Rajkumar, 2011; Clark, 2007). Temporary or permanent hemodialysis is indicated if the patient’s GFR is critically low and if symptomatic uremia becomes an issue. The use of high-cutoff dialyzers may effectively remove serum free light chains and can lead to improved renal function (Finkel, Cohen, Shirali, & Abudayyeh, 2016; Hutchison et al., 2007, 2009) or independence from hemodialysis among patients receiving a bortezomib-based regimen (Bridoux et al., 2016).
Evidence-Based Recommendations for Treatment of Renal Injury
Level of Evidence I
• Prompt initiation of effective MM therapy is recommended. This is critical for long-term kidney health.
• In line with the International Myeloma Working Group (IMWG) recommendations for treating patients with renal injury with bortezomib-based regimens (Dimopoulos, Sonneveld, et al., 2016), the IMF NLB recommends bortezomib as the cornerstone in the management of patients with renal injury.
Level of Evidence I-III
• Prompt treatment using classes of drugs approved by the U.S. Food and Drug Administration (FDA) is recommended to treat patients with relapsed or refractory myeloma and renal injury. The FDA-approved drugs that can be used for patients with simultaneous renal injury and active MM include immunomodulatory agents such as thalidomide (level I [Picot, Cooper, Bryant, & Clegg, 2011]); lenalidomide and pomalidomide (level III [Celgene, 2016, 2017a]); proteasome inhibitors such as bortezomib (level I [Picot et al., 2011]), carfilzomib (level III [Amgen Inc., 2016a]), and ixazomib (level III [Takeda Oncology, 2016]); and monoclonal antibodies such as elotuzumab (level III [Bristol-Myers Squibb, 2015]), and daratumumab (level III [Janssen Biotech, 2017]).
Evidence-Based Recommendations for Bone Health in Patients With Kidney Disease
Level of Evidence I
• Bisphosphonate therapy is recommended to prevent skeletal-related events, but not in patients with GFR of less than 30 ml per minute (Terpos, Roodman, & Dimopoulos, 2013).
• Bisphosphonates, such as zoledronic acid and pamidronate, are potent inhibitors of bone resorption that promote bone formation. However, bisphosphonate use in patients with renal injury or CKD may be toxic to the kidneys (Berenson, 1997). In patients with grade 1 or 2 CKD, the serum creatinine level should be monitored and the GFR should be estimated using the CKD-EPI calculator prior to each dose, as well as periodic monitoring with 24-hour urine to assess for albuminuria (NIDDKD, 2016).
• For patients who are taking bisphosphonates, it is recommended that they also take calcium 1,000 mg per day and vitamin D 400 IU per day. However, calcium supplementation remains contraindicated in patients with hypercalcemia (Faiman et al., 2011; Miceli et al., 2011).
Level of Evidence III
• The use of ESAs is recommended. Anemia, defined in patients with MM as a hemoglobin value 2 g/dl below the institutional limits of normal, often is present in patients with moderate-to-severe renal dysfunction. The use of ESAs in renal disease is common practice related to decreased renal production of erythropoietin. The use of ESAs to manage chemotherapy-induced anemia remains controversial because of concerns related to shortened survival seen in other cancers. An increased risk of stroke and thromboembolic events can be observed in patients with MM on ESA for prophylactic use (Anaissie et al., 2012. Interventions that minimize this risk should be initiated prior to use of ESA, such as employing general venous thromboembolic event precautions (ambulation, antiplatelet, and anticoagulation treatment when appropriate). According to the NCCN (2016d), CKD is not a contraindication for receiving ESAs. Obtaining baseline serum iron, ferritin, and serum total iron binding capacity are recommended prior to initiating oral iron therapy (NCCN, 2017c).
Evidence-Based Recommendations for Monitoring Multiple Myeloma and Renal Function
Level of Evidence V
• Close monitoring of a patient’s MM disease burden and renal injury is recommended by ensuring that the patient obtain routine blood chemistry, CBC, and myeloma-specific biomarker tests as part of every clinic visit (Faiman et al., 2017; Faiman, Mangan, Spong, & Tariman, 2011; Kurtin et al., 2016).
• Laboratory monitoring should be performed monthly or based on institutional routine clinic follow-up for stable disease (Kurtin et al., 2016). Myeloma-specific biomarkers should include serum and urine protein electrophoresis, quantitative immunoglobulins, 24-hour urine for protein electrophoresis, and serum free light chain assay (Kurtin, Bertolotti, Brigle, & Verina, 2016). The frequency of laboratory testing may change depending on the degree of renal failure and the patient’s response to therapy (Dimopoulos, Sonneveld, et al., 2016; Kurtin et al., 2016).
Evidence-Based Recommendations: Patient Education About Multiple Myeloma Oncology Emergencies
Level of Evidence V
• Patients and their caregivers should be taught about the additional burden of comorbid conditions on renal function. The National Kidney Foundation identifies risk factors for CRD, including diabetes, cardiovascular disease, hypertension, age (older than 60 years), U.S. racial or ethnic minority status, and family history of CKD (Levey et al., 2013).
• Comorbid conditions should be monitored closely and managed effectively in partnership with patients, caregivers, and nephrologists to avoid any additional injury to the kidney. Patients with MM are at increased risk for renal failure, not only from MM but also from aging itself (Palumbo et al., 2014). Diabetes and hypertension are the leading causes of end-stage renal disease, and diabetes mellitus is the number one cause of kidney failure. Almost half of all new patients on dialysis have diabetes, making it the fastest growing risk factor for kidney disease. Uncontrolled hypertension is both a cause and complication of CKD and should be carefully treated and controlled in all patients (Song, Cong, & Wilson, 2016).
Level of Evidence VI
• Oncology clinicians should address weight control, exercise, smoking cessation, and medications for adequate blood pressure or blood sugar control. Adequate health maintenance and preventive screenings for hypertension, diabetes, obesity, and smoking-related health conditions may prevent or slow the progression to kidney failure (Faiman et al., 2011; Tariman et al., 2016).
Conclusion
Patients with MM are living longer through advances in treatment and supportive measures. Nurses play pivotal roles in the education about, assessment of, and early intervention for OEs that can lead to mortality and morbidity in the myeloma survivor. Implementing evidence-based recommendations can reduce morbidity and mortality and increase patient QOL.
Hematologic toxicity is an expected but manageable side effect of the novel therapies that are used to treat MM. A combination of patient and caregiver education, careful monitoring of the patient’s CBC, and a comprehensive patient review of systems are all necessary to aid in the prevention of serious side effects. Early intervention is often the key to promoting patient adherence to therapeutic regimens and enhancing patient outcomes.
The authors gratefully acknowledge Rafat Abonour, MD, Brian G.M. Durie, MD, and Diane P. Moran, RN, MA, EdM, at the International Myeloma Foundation for their review of this manuscript.
About the Author(s)
Kevin Brigle, PhD, NP, is an oncology nurse practitioner in the Massey Cancer Center at Virginia Commonwealth University in Richmond; Amy Pierre, ANP-BC, is a nurse practitioner at Hackensack Meridian Health in New Jersey; Elizabeth Finley-Oliver, BSN, RN, OCN®, is a hematology RN at the Moffitt Cancer Center in Tampa, FL; Beth Faiman, PhD, MSN, APRN-BC, AOCN®, is a nurse practitioner in the Department of Hematology and Medical Oncology at the Cleveland Clinic Taussig Cancer Institute in Ohio; Joseph D. Tariman, PhD, RN, ANP-BC, FAAN, is an assistant professor in the School of Nursing at DePaul University in Chicago, IL; and Teresa Miceli, BSN, RN, OCN®, is a BMT RN care coordinator at the Mayo Clinic in Minneapolis, MN. The authors take full responsibility for this content. This supplement was supported by the International Myeloma Foundation, with funding from Celgene Corporation, Karyopharm Therapeutics, and Takeda Oncology. Writing and editorial support was provided by Eubio Medical Communications. Brigle is an independent contractor for Amgen, Celgene Corporation, Janssen Pharmaceuticals, Seattle Genetics, and Takeda Oncology. Pierre has served on speakers bureaus for Amgen and Celgene Cororation. Finley-Oliver has previously consulted and served on speakers bureaus for Amgen, Celgene Corporation, and Clinical Care Options. Faiman consults and serves on speakers bureaus for Amgen, Bristol-Myers Squibb, Celgene Corporation, and Takeda Oncology, and has received support from Celgene Corporation and Takeda Oncology. The article has been reviewed by independent peer reviewers to ensure that it is objective and free from bias. Brigle can be reached at kbrigle@vcu.edu, with copy to CJONEditor@ons.org. (Submitted June 2017. Accepted July 27, 2017.)
References
Amgen Inc. (2016a). Kyprolis® (carfilzomib) [Package insert]. Retrieved from http://pi.amgen.com/~/media/amgen/repositorysites/pi-amgen-com/kyprolis…
Amgen Inc. (2016b). Xgeva® (denosumab) [Package insert]. Retrieved from http://pi.amgen.com/united_states/xgeva/xgeva_pi.pdf
Anaissie, E.J., Coleman, E.A., Goodwin, J.A., Kennedy, R.L., Lockhart, K.D., Stewart, C.B., . . . Barlogie, B. (2012). Prophylactic recombinant erythropoietin therapy and thalidomide are predictors of venous thromboembolism in patients with multiple myeloma: Limited effectiveness of thromboprophylaxis. Cancer, 118, 549–557. https://doi.org/10.1002/cncr.26302
Aster, R.H., & Bougie, D.W. (2007). Drug-induced immune thrombocytopenia. New England Journal of Medicine, 357, 580–587. https://doi.org/10.1056/NEJMra066469
Avvisati, G., Tirindelli, M.C., & Annibali, O. (2003). Thrombocytopenia and hemorrhagic risk in cancer patients. Critical Reviews in Oncology/Hematology, 48(Suppl.), S13–S16.
Berenson, J.R., Rosen, L., Vescio, R., Lau, H.S., Woo, M., Sioufi, A., . . . Seaman, J.J. (1997). Pharmacokinetics of pamidronate disodium in patients with cancer with normal or impaired renal function. Journal of Clinical Pharmacology, 37, 285–290.
Birgegård, G., Gascón, P., & Ludwig, H. (2006). Evaluation of anaemia in patients with multiple myeloma and lymphoma: Findings of the European Cancer Anaemia Survey. European Journal of Haematology, 77, 378–386. https://doi.org/10.1111/j.1600-0609.2006.00739.x
Blimark, C., Holmberg, E., Mellqvist, U.H., Landgren, O., Björkholm, M., Hultcrantz, M., . . . Kristinsson, S.Y. (2015). Multiple myeloma and infections: A population-based study on 9253 multiple myeloma patients. Haematologica, 100, 107–113. https://doi.org/10.3324/haematol.2014.107714
Bodey, G.P., Buckley, M., Sathe, Y.S., & Freireich, E.J. (1966). Quantitative relationships between circulating leukocytes and infection in patients with acute leukemia. Annals of Internal Medicine, 64, 328–340. https://doi.org/10.7326/0003-4819-64-2-328
Bridoux, F., Pegourie, B., Augeul-Meunier, K., Royer, B., Joly, B., Lamy, T., . . . Fermand, J-P. (2016). Treatment of myeloma cast nephropathy (MCN): A randomized trial comparing intensive haemodialysis (HD) with high cut-off (HCO) or standard high-flux dialyzer in patients receiving a bortezomib-based regimen. Blood, 128, 978.
Bristol-Myers Squibb. (2015). Empliciti® (elotuzumab) [Package insert]. Retrieved from http://packageinserts.bms.com/pi/pi_empliciti.pdf
Burnette, B.L., Leung, N., & Rajkumar, S.V. (2011). Renal improvement in myeloma with bortezomib plus plasma exchange. New England Journal of Medicine, 364, 2365–2366. https://doi.org/10.1056/NEJMc1101834
Celgene. (2016). Pomalyst® (pomalidomide) [Package insert]. Retrieved from http://www.celgene.com/content/uploads/pomalyst-pi.pdf
Celgene. (2017a). Revlimid® (lenalidomide) [package insert]. Retrieved from http://www.celgene.com/content/uploads/revlimid-pi.pdf
Celgene. (2017b). Thalomid® (thalidomide) [Package insert]. Retrieved from http://www.celgene.com/content/uploads/thalomid-pi.pdf
Centers for Disease Control and Prevention. (2017). Cancer, infection, and sepsis fact sheet. Retrieved from https://www.cdc.gov/sepsis/pdfs/cancer-infection-and-sepsis-fact-sheet…
Clark, W.F. (2007). Correction: Plasma exchange when myeloma presents as acute renal failure. Annals of Internal Medicine, 146, 471. https://doi.org/10.7326/0003-4819-146-6-2007 03200-00024
Clemente, C.D. (1985). The back, vertebral column, and spinal cord. In, Anatomy: A regional atlas of the human body (3rd ed., pp. 517–573). Baltimore, MD: Urban and Schwarzenberg.
Dammacco, F., Silvestris, F., Castoldi, G.L., Grassi, B., Bernasconi, C., Nadali, G., . . . Resegotti, L. (1998). The effectiveness and tolerability of epoetin alfa in patients with multiple myeloma refractory to chemotherapy. International Journal of Clinical and Laboratory Research, 28, 127–134.
Damron, B.H., Brant, J.M., Belansky, H.B., Friend, P.J., Samsonow, S., & Schaal, A. (2009). Putting Evidence Into Practice: Prevention and management of bleeding in patients with cancer. Clinical Journal of Oncology Nursing, 13, 573–583. https://doi.org/10.1188/09.CJON.573-583
Dimopoulos, M.A., Cheung, M.C., Roussel, M., Liu, T., Gamberi, B., Kolb, B., . . . Facon, T. (2016). Impact of renal impairment on outcomes with lenalidomide and dexamethasone treatment in the FIRST trial, a randomized, open-label phase 3 trial in transplant-ineligible patients with multiple myeloma. Haematologica, 101, 363–370. https://doi.org/10.3324/haematol.2015.133629
Dimopoulos, M.A., Hillengass, J., Usmani, S., Zamagni, E., Lentzsch, S., Davies, F.E., . . . Terpos, E. (2015). Role of magnetic resonance imaging in the management of patients with multiple myeloma: A consensus statement. Journal of Clinical Oncology, 33, 657–664. https://doi.org/10.1200/JCO.2014.57.9961
Dimopoulos, M.A., Palumbo, A., Corradini, P., Cavo, M., Delforge, M., Di Raimondo, F., . . . Moreau, P. (2016). Safety and efficacy of pomalidomide plus low-dose dexamethasone in STRATUS (MM-010): A phase 3b study in refractory multiple myeloma. Blood, 128, 497–503. https://doi.org/10.1182/blood-2016-02-700872
Dimopoulos, M.A., Sonneveld, P., Leung, N., Merlini, G., Ludwig, H., Kastritis, E., . . . Terpos, E. (2016). International Myeloma Working Group recommendations for the diagnosis and management of myeloma-related renal impairment. Journal of Clinical Oncology, 34, 1544–1557. https://doi.org/10.1200/jco.2015.65.0044
Dimopoulos, M.A., Swern, A.S., Li, J.S., Hussein, M., Weiss, L., Nagarwala, Y., & Baz, R. (2014). Efficacy and safety of long-term treatment with lenalidomide and dexamethasone in patients with relapsed/refractory multiple myeloma. Blood Cancer Journal, 4, e257. https://doi.org/10.1038/bcj.2014.77
Dispenzieri, A., & Kyle, R.A. (2005). Neurological aspects of multiple myeloma and related disorders. Best Practice and Research Clinical Haematology, 18, 673–688. https://doi.org/10.1016/j.beha.2005.01.024
Doshi, M., Lahoti, A., Danesh, F.R., Batuman, V., & Sanders, P.W. (2016). Paraprotein-related kidney disease: Kidney injury from paraproteins—What determines the site of injury? Clinical Journal of the American Society of Nephrology, 11, 2288–2294. https://doi.org/10.2215/cjn.02560316
Faiman, B., & Bilotti, E. (2014). Multiple myeloma. In M. Olsen & L.J. Zitella (Eds.), Hematologic malignancies in adults (pp. 445–498). Pittsburgh, PA: Oncology Nursing Society.
Faiman, B., Doss, D., Colson, K., Mangan, P., King, T., Tariman, J., & IMF NLB. (2017). Renal, GI, and peripheral nerves: Evidence-based recommendations for the management of symptoms and care for patients with multiple myeloma. Clinical Journal of Oncology Nursing, 21(Suppl., 5), 19–36.
Faiman, B.M., Mangan, P., Spong, J., & Tariman, J.D. (2011). Renal complications in multiple myeloma and related disorders: Survivorship care plan of the International Myeloma Foundation Nurse Leadership Board. Clinical Journal of Oncology Nursing, 15(Suppl.), S66–S76. https://doi.org/10.1188/11.CJON.S1.66-76
Finkel, K.W., Cohen, E.P., Shirali, A., & Abudayyeh, A. (2016). Paraprotein-related kidney disease: Evaluation and treatment of myeloma cast nephropathy. Clinical Journal of the American Society of Nephrology, 11, 2273–2279. https://doi.org/10.2215/cjn.01640216
Fliser, D., Laville, M., Covic, A., Fouque, D., Vanholder, R., Juillard, L., & Van Biesen, W. (2012). A European Renal Best Practice (ERBP) position statement on the Kidney Disease Improving Global Outcomes (KDIGO) clinical practice guidelines on acute kidney injury: Part 1: Definitions, conservative management and contrast-induced nephropathy. Nephrology, Dialysis, Transplantation, 27, 4263–4272. https://doi.org/10.1093/ndt/gfs375
Flowers, C.R., & Karten, C. (2013). Communicating safe outpatient management of fever and neutropenia. Journal of Oncology Practice, 9(4), 207–210. https://doi.org/10.1200/JOP.2012.000815
Flowers, C.R., Seidenfeld, J., Bow, E.J., Karten, C., Gleason, C., Hawley, D.K., . . . Ramsey, S.D. (2013). Antimicrobial prophylaxis and outpatient management of fever and neutropenia in adults treated for malignancy: American Society of Clinical Oncology clinical practice guideline. Journal of Clinical Oncology, 31, 794–810. https://doi.org/10.1200/JCO.2012.45.8661
Focus Information Technology. (2017). Corrected calcium calculator. Retrieved from http://www.perinatology.com/calculators/Corrected%20Calcium.htm
Freifeld, A.G., Bow, E.J., Sepkowitz, K.A., Boeckh, M.J., Ito, J.I., Mullen, C.A., . . . Wingard, J.R. (2011). Clinical practice guideline for the use of antimicrobial agents in neutropenic patients with cancer: 2010 update by the Infectious Diseases Society of America. Clinical Infectious Diseases, 52, 427–431. https://doi.org/10.1093/cid/ciq147
Galson, D.L., Silbermann, R., & Roodman, G.D. (2012). Mechanisms of multiple myeloma bone disease. BoneKEy Reports, 1, 135. https://doi.org/10.1038/bonekey.2012.135
Gavriatopoulou, M., Terpos, E., Kastritis, E., & Dimopoulos, M.A. (2016). Current treatments for renal failure due to multiple myeloma. Expert Opinion on Pharmacotherapy, 17, 2165–2177.
Greipp, P.R., San Miguel, J., Durie, B.G., Crowley, J.J., Barlogie, B., Bladé, J., . . . Westin, J. (2005). International staging system for multiple myeloma. Journal of Clinical Oncology, 23, 3412–3420. https://doi.org/10.1200/JCO.2005.04.242
Ha, K.Y., Kim, Y.H., & Kim, H.W. (2013). Multiple myeloma and epidural spinal cord compression: Case presentation and a spine surgeon’s perspective. Journal of Korean Neurosurgical Society, 54, 151–154. https://doi.org/10.3340/jkns.2013.54.2.151
Hsu, R.K., & Hsu, C.Y. (2016). The role of acute kidney injury in chronic kidney disease. Seminars in Nephrology, 36, 283–292. https://doi.org/10.1016/j.semnephrol.2016.05.005
Hughes, W.T., Armstrong, D., Bodey, G.P., Bow, E.J., Brown, A.E., Calandra, T., . . . Young, L.S. (2002). 2002 guidelines for the use of antimicrobial agents in neutropenic patients with cancer. Clinical Infectious Diseases, 34, 730–751. https://doi.org/10.1086/339215
Hussein, M.A., Vrionis, F.D., Allison, R., Berenson, J., Berven, S., Erdem, E., . . . Durie, B.G. (2008). The role of vertebral augmentation in multiple myeloma: International Myeloma Working Group consensus statement. Leukemia, 22, 1479–1484. https://doi.org/10.1038/leu.2008.127
Hutchison, C.A., Bradwell, A.R., Cook, M., Basnayake, K., Basu, S., Harding, S., . . . Cockwell, P. (2009). Treatment of acute renal failure secondary to multiple myeloma with chemotherapy and extended high cut-off hemodialysis. Clinical Journal of the American Society of Nephrology, 4, 745–754. https://doi.org/10.2215/cjn.04590908
Hutchison, C.A., Cockwell, P., Reid, S., Chandler, K., Mead, G.P., Harrison, J., . . . Bradwell, A.R. (2007). Efficient removal of immunoglobulin free light chains by hemodialysis for multiple myeloma: In vitro and in vivo studies. Journal of the American Society of Nephrology, 18, 886–895. https://doi.org/10.1681/asn.2006080821
Janssen Biotech. (2017). Darzalex® (daratumumab) [Package insert]. Retrieved from https://www.darzalexhcp.com/shared/product/darzalex/darzalex-prescribin…
Kaplan, M. (2012a). Hypercalcemia of malignancy. In M. Kaplan (Ed.), Understanding and managing oncologic emergencies: A resource for nurses (2nd ed., pp 103–156). Pittsburgh, PA: Oncology Nursing Society.
Kaplan, M. (2012b). Spinal cord compression. In M. Kaplan (Ed.), Understanding and managing oncologic emergencies: A resource for nurses (2nd ed., pp 337–384). Pittsburgh, PA: Oncology Nursing Society.
King, T., & Faiman, B. (2017). Steroid-associated side effects: A symptom management update on multiple myeloma. Clinical Journal of Oncology Nursing, 21, 240–249. https://doi.org/10.1188/17.CJON.240-249
Kyle, R.A. (1975). Multiple myeloma: Review of 869 cases. Mayo Clinic Proceedings, 50, 29–40.
Kyle, R.A., Gertz, M.A., Witzig, T.E., Lust, J.A., Lacy, M.Q., Dispenzieri, A., . . . Greipp, P.R. (2003). Review of 1027 patients with newly diagnosed multiple myeloma. Mayo Clinic Proceedings, 78, 21–33. https://doi.org/10.4065/78.1.21
Kurtin, S., Bertolotti, P., Brigle, K., & Verina, D. (2016). Updates in the diagnosis and monitoring of multiple myeloma. Journal of the Advanced Practitioner in Oncology, 7, 59–70. https://doi.org/10.6004/jadpro.2016.7.2.15
Laubach, J., Garderet, L., Mahindra, A., Gahrton, G., Caers, J., Sezer, O., . . . Richardson, P.G. (2016). Management of relapsed multiple myeloma: Recommendations of the International Myeloma Working Group. Leukemia, 30, 1005–1017. https://doi.org/10.1038/leu.2015.356
Levey, A.S., Coresh, J., Balk, E., Kausz, A.T., Levin, A., Steffes, M.W., . . . Eknoyan, G. (2013). National Kidney Foundation practice guidelines for chronic kidney disease: Evaluation, classification, and stratification. Annals of Internal Medicine, 139, 137–147.
Lewis, M.A., Hendrickson, A.W., & Moynihan, T.J. (2011). Oncologic emergencies: Pathophysiology, presentation, diagnosis, and treatment. CA: A Cancer Journal for Clinicians, 61, 287–314. https://doi.org/10.3322/caac.20124
Maes, K., Nemeth, E., Roodman, G.D., Huston, A., Esteve, F., Freytes, C., . . . Ganz, T. (2010). In anemia of multiple myeloma, hepcidin is induced by increased bone morphogenetic protein 2. Blood, 116, 3635–3644. https://doi.org/10.1182/blood-2010-03-274571
Miceli, T., Colson, K., Gavino, M., & Lilleby, K. (2008). Myelosuppression associated with novel therapies in patients with multiple myeloma: Consensus statement of the IMF Nurse Leadership Board. Clinical Journal of Oncology Nursing, 12(Suppl. 3), S13–S20. https://doi.org/10.1188/08.CJON.S1.13-19
Miceli, T.S., Colson, K., Faiman, B.M., Miller, K., & Tariman J.D. (2011). Maintaining bone health in patients with multiple myeloma: Survivorship care plan of the International Myeloma Foundation Nurse Leadership Board. Clinical Journal of Oncology Nursing, 15(Suppl.), S9–S23. https://doi.org/10.1188/11.S1.CJON.9-23
Millennium Pharmaceuticals. (2017). Velcade® (bortezomib) [Package insert]. Retrieved from http://www.velcade.com/files/PDFs/VELCADE_PRESCRIBING_INFORMATION.pdf
Miller. (2010). Anemia. In C. Brown (Ed.), Guide to oncology symptom management (pp. 29–47). Pittsburgh, PA: Oncology Nursing Society.
Moos, S.I., van Vemde, D.N., Stoker, J., & Bipat, S. (2013). Contrast induced nephropathy in patients undergoing intravenous (IV) contrast enhanced computed tomography (CECT) and the relationship with risk factors: A meta-analysis. European Journal of Radiology, 82(9), e387–e399. https://doi.org/10.1016/j.ejrad.2013.04.029
National Cancer Institute. (2010). Common terminology criteria for adverse events [v.4.03]. Retrieved from https://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03_2010-06-14_QuickReference…
National Cancer Institute. (2017). NCI dictionary of cancer terms: Myelosuppression. Retrieved from https://www.cancer.gov/publications/dictionaries/cancer-terms?cdrid=441…
National Comprehensive Cancer Network. (2016). NCCN Guidelines for Patients®: Multiple myeloma [v.1.2016]. Retrieved from https://www.nccn.org/patients/guidelines/content/PDF/myeloma.pdf
National Comprehensive Cancer Network. (2017a). NCCN Clinical Practice Guidelines in Oncology: Cancer- and chemotherapy-induced anemia [v.1.2017]. Retrieved from https://www.nccn.org/professionals/physician_gls/pdf/anemia.pdf
National Comprehensive Cancer Network. (2017b). NCCN Clinical Practice Guidelines in Oncology: Multiple myeloma [v.3.2017]. Retrieved from https://www.nccn.org/professionals/physician_gls/pdf/myeloma.pdf
National Comprehensive Cancer Network. (2017c). NCCN Clinical Practice Guidelines in Oncology: Prevention and treatment of cancer-related infections [v.2.2017]. Retrieved from https://www.nccn.org/professionals/physician_gls/pdf/infections.pdf
National Institute of Diabetes and Digestive and Kidney Diseases. (2016). CKD-EPI adults (conventional units). Retrieved from https://www.niddk.nih.gov/health-information/health-communication-progr… tional-unit-ckd-epi/Pages/default.aspx
National Institute of Diabetes and Digestive and Kidney Diseases. (2017). Anemia in chronic kidney disease. Retrieved from https://www.niddk.nih.gov/health-information/kidney-disease/chronic-kid…
Niesvizky, R., & Badros, A.Z. (2010). Complications of multiple myeloma therapy, part 2: Risk reduction and management of venous thromboembolism, osteonecrosis of the jaw, renal complications, and anemia. Journal of the National Comprehensive Cancer Network, 8(Suppl. 1), S13–S20.
Noonan, K., Rome, S., Faiman, B., Verina, D., & the IMF NLB. (2017). Heart and lung complications: Assessment and prevention of venous thromboembolism and cardiovascular disease in patients with multiple myeloma. Clinical Journal of Oncology Nursing, 21(Suppl. 5), 37–46.
Novartis. (2016). Farydak® (panobinostat) [Package insert]. Retrieved from https://www.pharma.us.novartis.com/sites/www.pharma.us.novartis.com/fil…
O’Grady, N.P., Alexander, M., Burns, L.A., Dellinger, E.P., Garland, J., Heard, S.O., . . . Saint, S. (2011). Guidelines for the prevention of intravascular catheter-related infections. Clinical Infectious Diseases, 52(9), e162–e193. https://doi.org/10.1093/cid/cir257
Pace, A.F. (2015). Electrolyte imbalances, syndrome of inappropriate antidiuretic hormone, and tumor lysis syndrome. In C. Brown (Ed.), A guide to oncology symptom management (2nd ed., pp 319–368). Pittsburgh, PA: Oncology Nursing Society.
Palumbo, A., Rajkumar, S.V., San Miguel, J.F., Larocca, A., Niesvizky, R., Morgan, G., . . . Orlowski, R.Z. (2014). International Myeloma Working Group consensus statement for the management, treatment, and supportive care of patients with myeloma not eligible for standard autologous stem-cell transplantation. Journal of Clinical Oncology, 32, 587–600. https://doi.org/10.1200/JCO.2013.48.7934
Picot, J., Cooper, K., Bryant, J., & Clegg, A.J. (2011). The clinical effectiveness and cost-effectiveness of bortezomib and thalidomide in combination regimens with an alkylating agent and a corticosteroid for the first-line treatment of multiple myeloma: A systematic review and economic evaluation. Health Technology Assessment, 15(41), 1–204. https://doi.org/10.3310/hta15410
Rajkumar, S.V., & Kumar, S. (2016). Multiple myeloma: Diagnosis and treatment. Mayo Clinic Proceedings, 91, 101–119. https://doi.org/10.1016/j.mayocp.2015.11.007
Richardson, P., Schlag, R., Khuageva, N., Dimopoulos, M., Shpilberg, O., Kropff, M., . . . San Miguel, J.F. (2011). Characterization of haematological parameters with bortezomib-melphalan-prednisone versus melphalan-prednisone in newly diagnosed myeloma, with evaluation of long-term outcomes and risk of thromboembolic events with use of erythropoiesis-stimulating agents: Analysis of the VISTA trial. British Journal of Haematology, 153, 212–221. https://doi.org/10.1111/j.1365-2141.2011.08569.x
Rizzo, J.D., Brouwers, M., Hurley, P., Seidenfeld, J., Arcasoy, M.O., Spivak, J.L., . . . Somerfield, M.R. (2010). American Society of Hematology/American Society of Clinical Oncology clinical practice guideline update on the use of epoetin and darbepoetin in adult patients with cancer. Blood, 116, 4045–4059. https://doi.org/10.1182/blood-2010-08-300541
Rome, S., Noonan, K., Bertolotti, P., Tariman, J.D., Miceli, T., & IMF NLB. (2017). Bone health, pain, and mobility: Evidence-based recommendations for patients with multiple myeloma. Clinical Journal of Oncology Nursing, 21(Suppl., 5), 47–59.
Roodman, G.D. (2008). Skeletal imaging and management of bone disease. Hematology: American Society of Hematology Education Program, 2008, 313–319. https://doi.org/10.1182/asheducation-2008.1.313
Schiffer, C.A., Anderson, K.C., Bennett, C.L., Bernstein, S., Elting, L.S., Goldsmith, M., . . . Wagnon, A.H. (2001). Platelet transfusion for patients with cancer: Clinical practice guidelines of the American Society of Clinical Oncology. Journal of Clinical Oncology, 19, 1519–1538. https://doi.org/10.1200/JCO.2001.19.5.1519
Schiffer, C.A., Mangu, P.B., Wade, J.C., Camp-Sorrell, D., Cope, D.G., El-Rayes, B.F., . . . Levine, M. (2013). Central venous catheter care for the patient with cancer: American Society of Clinical Oncology clinical practice guideline. Journal of Clinical Oncology, 31, 1357–1370. https://doi.org/10.1200/JCO.2012.45.5733
Shane, E., & Berenson, J.R. (2015). Treatment of hypercalcemia. UpToDate. Retrieved from http://www.uptodate.com/contents/treatment-of-hypercalcemia
Smith, T.J., Bohlke, K., Lyman, G.H., Carson, K.R., Crawford, J., Cross, S.J., . . . Armitage, J.O. (2015). Recommendations for the use of WBC growth factors: American Society of Clinical Oncology clinical practice guideline update. Journal of Clinical Oncology, 33, 3199–3212. https://doi.org/10.1200/JCO.2015.62.3488
Snowden, J.A., Ahmedzai, S.H., Ashcroft, J., D’Sa, S., Littlewood, T., Low, E., . . . Bird, J.M. (2011). Guidelines for supportive care in multiple myeloma 2011. British Journal of Haematology, 154, 76–103. https://doi.org/10.1111/j.1365-2141.2011.08574.x
Song, X., Cong, Z., & Wilson, K. (2016). Real-world treatment patterns, comorbidities, and disease-related complications in patients with multiple myeloma in the United States. Current Medical Research and Opinion, 32, 95–103. https://doi.org/10.1185/03007995.2015.1105202
Sternlicht, H., & Glezerman, I.G. (2015). Hypercalcemia of malignancy and new treatment options. Therapeutics and Clinical Risk Management, 11, 1779–1788.
Takeda Oncology. (2016). Ninlaro® (ixazomib) [Package insert]. Retrieved from https://www.ninlarohcp.com/pdf/prescribing-information.pdf
Tariman, J.D., Gleason, C., Faiman, B., Doss, D., Catamero, D., Bishop-Royse, J., . . . Moran, D. (2016). Lack of health maintenance examinations and risk in myeloma patients. Cancer Medicine, 5, 1425–1435. https://doi.org/10.1002/cam4.716
Teh, B.W., Harrison, S.J., Pellegrini, M., Thursky, K.A., Worth, L.J., & Slavin, M.A. (2014). Changing treatment paradigms for patients with plasma cell myeloma: Impact upon immune determinants of infection. Blood Reviews, 28, 75–86. https://doi.org/10.1016/j.blre.2014.01.004
Teh, B.W., Harrison, S.J., Worth, L.J., Spelman, T., Thursky, K.A., & Slavin, M.A. (2015). Risks, severity and timing of infections in patients with multiple myeloma: A longitudinal cohort study in the era of immunomodulatory drug therapy. British Journal of Haematology, 171, 100–108. https://doi.org/10.1111/bjh.13532
Terpos, E., Christoulas, D., Kastritis, E., Katodritou, E., Pouli, A., Michalis, E., . . . Dimopoulos, M.A. (2013). The Chronic Kidney Disease Epidemiology Collaboration cystatin C (CKD-EPI-CysC) equation has an independent prognostic value for overall survival in newly diagnosed patients with symptomatic multiple myeloma: Is it time to change from MDRD to CKD-EPI-CysC equations? European Journal of Haematology, 91, 347–355.
Terpos, E., Dimopoulos, M.A., & Moulopoulos, L.A. (2016). The role of imaging in the treatment of patients with multiple myeloma in 2016. American Society of Clinical Oncology Educational Book, 35, e407–e417. https://doi.org/10.14694/EDBK_159074
Terpos, E., Kleber, M., Engelhardt, M., Zweegman, S., Gay, F., Kastritis, E., . . . Palumbo, A. (2015). European Myeloma Network guidelines for the management of multiple myeloma-related complications. Haematologica, 100, 1254–1266.
Terpos, E., Morgan, G., Dimopoulos, M.A., Drake, M.T., Lentzsch, S., Raje, N., . . . Roodman, G.D. (2013). International Myeloma Working Group recommendations for the treatment of multiple myeloma-related bone disease. Journal of Clinical Oncology, 31, 2347–2357. https://doi.org/10.1200/JCO.2012.47.7901
Terpos, E., Roodman, G.D., & Dimopoulos, M.A. (2013). Optimal use of bisphosphonates in patients with multiple myeloma. Blood, 121, 3325–3328. https://doi.org/10.1182/blood-2012-10-435750
Terpos, E., Christoulas, D., Kastritis, E., Bagratuni, T., Gavriatopoulou, M., Roussou, M., . . . Dimopoulos, M.A. (2016). High levels of periostin correlate with increased fracture rate, diffuse MRI pattern, abnormal bone remodeling and advanced disease stage in patients with newly diagnosed symptomatic multiple myeloma. Blood Cancer Journal, 6(10), e482. https://doi.org/10.1038/bcj.2016.90
Thomas, S.A., & Chung, S. (2016). Management of hypercalcemia of malignancy. Journal of Hematology Oncology Pharmacy, 6, 18–21.




